UK: Eyed Hawkmoth, F: Sphinx Demi-paon, D: Abendpfauenauge, RUS: Glazchatyi Brazhnik, S: Vide-Nattsvärmare; Videsvärmare, NL: Pauwoogpijlstaart, CZ: Zubokřídlec paví oko; Lišaj paví oko, H: esti pávaszem, E: esfinge ocelada, PL: Gryzuń pólpawik; Nastrosz pólpawik, FIN: Silmäkiitäjä; Sinisilmäkiitäjä, I: sfinge ocellata; sfinge dagli occhi, HR: okati ljiljak, DK: Aftenpåfugleøje, N: Kveldpåfugløye, EST: Silmiksuru.
Sphinx ocellata Linnaeus, 1758, Syst. Nat. (Edn 10) 1: 489.Type locality: Unspecified [Europe].
(Taxonomic note. The name Smerinthus is apparently derived from the Greek feminine noun 'Merinthos', but has been Latinised with the masculine -us ending and is thus, according to the ICZN article 30.1.3, masculine. As the ICZN rules that species names that are adjectives should agree in gender with the genus name, the common use of Smerinthus ocellata for this species is incorrect.)
[Further details on this species, as well as photos of all stages, can be found on Lepiforum.]

Holarctic; western Palaearctic region. Pleistocene refuge: Polycentric -- Pontomediterranean and Caspian refugia.



Wingspan: 70--95mm. Very similar in appearance to the other two Smerinthus species found in the western Palaearctic but distinguished by the presence of an apical thorn on the foretibia, and a large, circular hindwing ocellus. In addition to variations in forewing pattern intensity, differences in colour are also to be found on the hindwing, where pink may be replaced by yellow (f. flavescens Neumann); grey (f. pallida Tutt); or white (f. albescens Tutt). In f. rosea Bartel, the pink is abnormally deep and often coupled with yellowish brown forewings; in f. ollivryi Oberthür, the ocellus may be replaced by a buff brown patch.
There is also a certain degree of variation in wing outline and overall colour tone, particularly in males. The forewings can be narrower, elongated and falcate, with a more undulating distal margin. In others, particularly in the UK, the wings and body may have a pronounced violet-purple sheen. Some of these morphs can be more common in certain parts of Europe than in others, giving the impression of local races. This variation, coupled with naturally variable male genitalia, has led to several of these forms and morphs being described as good subspecies (e.g. Smerinthus ocellata cinerascens Staudinger, 1879) or even species (e.g. Smerinthus visinskasi Zolotuhin & Saldaitis, 2009).




A local species occurring year after year in a given area, while nearby localities, which may appear very suitable, never support a population. Even where present, numbers may fluctuate markedly from year to year due to the attentions of parasitic wasps, especially Microplitis ocellatae Bouché. Found in many different localities, especially disused railway cuttings overgrown with sallow (Salix spp.), riverine shingle bars, wet river valleys, country lanes bordered by willows (Salix spp.), apple orchards, coastal sand-hills, and suburban gardens. Up to 2000m in the Alps.
At rest, both sexes hang suspended amongst foliage, the cryptic forewing markings mimicking patterns on dead leaves, as with Mimas tiliae (Linnaeus, 1758). Its form is further broken up by the slight projection of the hindwings above the forewing costa. Also, as in Mimas tiliae, pairing takes place in such situations, usually after midnight, the male often hanging freely beneath the female until dusk the following day. After separation, females commence egg-laying almost immediately, continuing over four or five nights, with maximum activity between dusk and midnight. When alarmed, resting adults exhibit a very effective defence mechanism: the body is hunched as the forewings are flicked upwards to reveal a pair of glaring 'eyes' each side of a narrow, beak-like abdomen. Following more disturbance, this startling 'face' is further enhanced: the forewings are repeatedly lowered and raised, causing a blinking effect to the eyes. If this does not succeed, then refuge is sought on the ground. As a non-feeding species, flowers hold no attraction; light, however, can often lure large numbers of males.


Univoltine in northern latitudes, from the end of May to early July; farther south bivoltine, during April/May and again in August. Occasionally trivoltine, during April/May, June/July and August/September (Ganev, 1984). In the southern Urals, from mid June until mid July, with a partial second generation in early September (Nupponen & Fibiger, 2002).
OVUM: Oval, very shiny, pale green (1.6 x 1.4mm), becoming greyish white prior to hatching. Laid singly or in pairs beneath leaves not more than 2m above the ground.

LARVA: Full-fed: 70--80mm. Dimorphic: bluish green or apple-green.








On hatching, the young larva is about 5mm long, whitish green, with a pale pink horn. It clings tightly with all legs to a vein on the underside of a leaf. In the second instar, pale lateral stripes appear, the head becomes dorsally pointed and, when not feeding, it rests in a typical sphinx-like posture, holding on with the last three pairs of prolegs only. When so positioned beneath a leaf, the larva is very difficult to detect due to its excellent counter-shading. With growth, a number of changes take place: the horn gradually changes from pink, through purple, to blue in the final instar; the counter-shading becomes more noticeable; the dorsal surface becomes much paler than the ventral, with more of the prominent white or yellow tubercles; and the head eventually loses its pointed apex. Two colour forms are found: the commonest is bluish white with white lateral stripes and tubercles; the other, yellowish green with yellow markings and, occasionally, red blotches and stripe edging. Prior to pupation, all become suffused with brownish yellow before the larva wanders off to find a pupation site, which is usually some distance from the hostplant.

Up to 80 per cent of larvae in a colony may perish in any one year due to parasitoids, such as Microplitis ocellatae Bouché.
In northern regions found from early June until September, but in southern regions from early May until early October in two broods which often overlap.
Major Hostplants. Many species of Salix, Populus and Malus.
Minor Hostplants. Species of Betula, Alnus, Prunus, Pyrus, Tilia, ?Ligustrum and ?Viburnum. Amongst the Prunus complex, Prunus spinosa is frequently taken, whereas Prunus avium, Prunus padus, Prunus dulcis and Prunus persica are less favoured. In London, often found on the ornamental Prunus laurocerasus (cherry laurel). (The records for Ligustrum and Viburnum are suspect and may refer to Sphinx ligustri Linnaeus, 1758. Immature larvae of the latter look very similar to those of Smerinthus ocellatus.)
PUPA: 35--41mm. Rich, glossy blackish brown. Thickset, anteriorly blunted and tapering posteriorly, in shape resembling that of Laothoe populi (Linnaeus, 1758). Most are formed in an earthen cell 2--3 cm under the surface of the soil away from the hostplant. The overwintering stage.

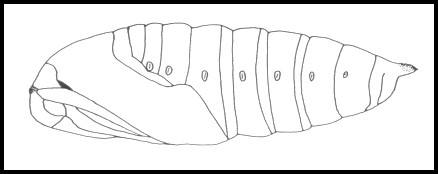
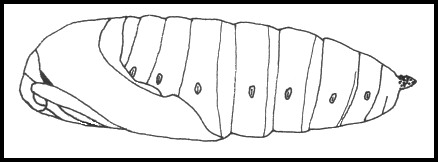
Ichneumonidae: Amblyteles armatorius (Förster, 1771), Banchus pictus Fabricius, 1798, Callajoppa cirrogaster (Schrank, 1781), Callajoppa exaltatoria (Panzer, 1804), Coelichneumon sinister (Wesmael, 1848), Cratichneumon sicarius (Gravenhorst, 1829), Diphyus palliatorius (Gravenhorst, 1829), Lymantrichneumon disparis (Poda, 1761), Netelia testacea (Gravenhorst, 1829), Netelia vinulae (Scopoli, 1763), Pimpla illecebrator (Villers, 1789), Protichneumon fusorius (Linnaeus, 1761), Protichneumon pisorius (Linnaeus, 1758), Pyramidophorus flavoguttatus Tischbein, 1882, Scolobates auriculatus (Fabricius, 1804); Eulophidae: Eulophus smerinthicida Bouček, 1959; Scelionidae: Telenomus punctatissimus (Ratzeburg, 1844); Braconidae: Cotesia abjecta (Marshall, 1885), Microplitis ocellatae Bouché, 1834, Microplitis deprimator (Fabricius, 1798), Microgaster tibialis Nees, 1834, Aleiodes praetor (Reinhard, 1863) [syn. Rogas praetor Reinhard, 1863]; Tachinidae: Blepharipa pratensis (Meigen, 1824), Compsilura concinnata (Meigen, 1824), Exorista grandis (Zetterstedt, 1844), Exorista sorbillans (Wiedemann, 1830), Frontina laeta (Meigen, 1824), Masicera sphingivora (Robineau-Desvoidy, 1830), Nilea innoxia (Robineau-Desvoidy, 1863), Phryxe erythrostoma (Hartig, 1838), Siphona cristata (Fabricius, 1805), Winthemia bohemanni (Zetterstedt, 1844), Winthemia cruentata (Rondani, 1859), Winthemia quadripustulata (Fabricius, 1794).



From western and southern Europe (including Portugal (Corley, 2004; Pires & Corley, 2007), Galicia (Pino Pérez et al., 2009; Fernández Vidal, 2016), Ireland (Carpenter, 1914) and Sicily (Mariani, 1939; Parenzan & Porcelli, 2006) eastward across the southern Urals (Eversmann, 1844; Derzhavets, 1984) to Novosibirsk and Krasnoyarsk (Izerskiy, 1999b), and western Mongolia (Khovd Province) (Grosser, 1982). Thence southwards to eastern Kazakhstan (Toropov, Milko, Zhdanko & Evdoshenko, 2023), the Altai (Zolotarenko, Petrova & Shiryaev, 1978; Izerskiy, 1999), Kyrgyzstan and western and northern Xinjiang Province (China). Also, Albania (Beshkov & Misja, 1995), the Republic of North Macedonia (Krpač et al., 2019), Greece, Turkey (de Freina, 1979; Ayberk & Akkuzu, 2005; Feza Doganlar, pers. comm.; de Freina, 2012; Okyar, 2012; Koçak & Kemal, 2018), Ukraine (Khalaim, 2022), Moldova (Tugulea & Tugulea, 2020), Armenia (Wąsala & Zamorski, 2015), the Republic of Georgia (Didmanidze, Petrov & Zolotuhin, 2013; Streltzov et al., 2022), Daghestan (Didmanidze, Petrov & Zolotuhin, 2013; Yakovlev et al., 2022), Azerbaijan (Snegovaya & Petrov, 2021), northern Iran (Sutton, 1963; Lehmann & Zahiri, 2011), southern Turkmenistan and Uzbekistan (Omonov et al., 2025). Absent from northern Scandinavia and Arctic Russia; however, recorded as far north as Emva in european Russia (Tatarinov, Sedykh & Dolgin, 2003).
On the Iberian Peninsula mainly confined to central (Zerny, 1927; Hurtado, Hiernaux & Pereira, 2011) and northern areas (Vallhonrat et al., 2004), but with isolated populations in the mountains of southern Spain and Portugal (Pérez De-Gregorio et al., 2001; Lara Ruiz, 2011; Garre, Rubio, Guerrero & Ortiz, 2020; Pedro Sandoval, iNaturalist 2023), even as far south as Cadiz (David Barros Cardona, iNaturalist 2018).
Extra-limital range. None.
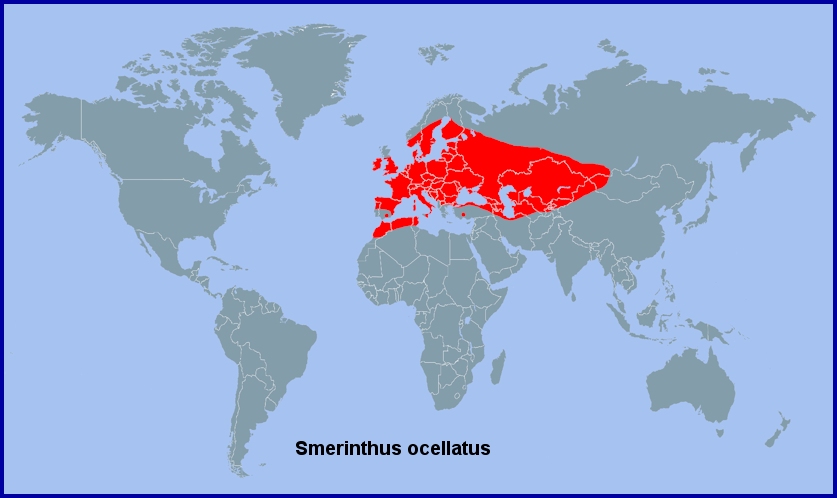
Northwest Africa, Sardinia and Corsica as Smerinthus ocellatus atlanticus Austaut, 1890.
 Return to species list
Return to species list