UK: Bat Hawkmoth; Dusky Hawkmoth; Mouse Hawkmoth, F: Sphinx Cendré; Sphinx chauve-souris; Sphinx vespertilio, D: Fledermausschwärmer, H: denevérszender, PL: Zmrocznik wieczorniak, FIN: Lepakkokiitäjä, I: Sfinge pipistrello, HR: sivokrili ljiljak.
Sphinx vespertilio Esper, 1780, Die Schmetterlinge 2: 178.Type locality: Verona, northern Italy.
[Further details on this species, as well as photos of all stages, can be found on Lepiforum.]
![A plate from Lucas, H. [1864]. A plate from Lucas, H. [1864].](lucas1864a.jpg)

Holarctic; western Palaearctic region. Pleistocene refuge: Poly/monocentric -- Adriatomediterranean and Pontomediterranean subsections of the Mediterranean refuge.

Wingspan: 60--80mm. Forewing almost uniform slate-grey; hindwing reddish pink, with indistinct dark markings, which distinguish Hyles vespertilio from other species of Hyles. A certain amount of variation is exhibited: in f. salmonea Oberthür there is no red on the hindwing; in f. flava Blachier the hindwing is yellow.



Unlike many members of this genus, Hyles vespertilio is not migratory and occurs in local, independent colonies up to 1700m altitude in Europe. In southern Bulgaria it is most common at around 1300m (Pittaway, pers. obs. 2009), but at higher altitudes in Asia Minor, i.e. 1700-2000m in the Caucasus (Shchurov, Poltavsky & Ilyina, 2013). Frequents well-drained, open, sunny, south-facing, gravel/scree slopes in warm, dry valleys. Hot shingle river-banks and bars are also a favourite haunt.



Whilst closely resembling Hyles euphorbiae (Linnaeus, 1758) in its feeding, mating and activity behaviour, adults rest mostly on the ground, usually amongst rocks and pebbles (in a river-bed, for instance), with which their coloration blends.
Over most of its range, May/June, with a partial-to-full second generation in August/September. At higher altitudes in central Europe and Bulgaria (Ganev, 1984) only one emergence may occur in some years, during June/July.
OVUM: Oval, slightly dorso-ventrally flattened (1.1 x 1.0mm), pale, glossy green. Laid singly or in pairs on the stems, leaves and flower-shoots of Epilobium, often with up to ten to a plant. Very occasionally found on stones next to a potential hostplant.

LARVA: Full-fed, 70--80mm.
The newly hatched larva is greenish-yellow, 3--4mm-long, with several longitudinal rows of fine black spots. With feeding the yellow is rapidly replaced by apple-green, upon which yellowish-white dorso-lateral and ventro-lateral lines appear, with the former being much more pronounced. There is no horn, or only a small tubercle where the horn would normally be.
In the second instar the pale longitudinal lines become creamy-yellow, with the green areas between them bearing numerous fine yellow dots, especially laterally. These are even more pronounced in the third instar, especially in the less common dark olive-green form. Both the dorso-lateral and ventro-lateral lines may be interupted on each segment by an orange spot; this is especially so in the dark form, where the basic olive-green colour gradually changes to dark brown.






When small, it feeds by day from beneath the leaf on which it rests, rarely leaving the hostplant until the fourth instar. At this stage the primary colour becomes greyish-brown, with a dorso-lateral line of greyish-yellow, orange-centred eye-spots. Large numbers of small, brown spots now cover the darker dorsal surface. The paler sides, between the dorso-lateral and ventro-lateral lines, bear numerous whitish-yellow spots and the ventral surface, legs, anal flap and head are dull pink. It now feeds only at night, hiding by day on the ground amongst small stones at the base of the hostplant. Many curl up in an excellent imitation of a pebble. When full grown in the final instar, the eye-spots and primary body colour are pale greyish-brown; very little change occurs in this grey coloration prior to pupation.




Most commonly found during June, July and September, often in considerable numbers over a small area; however, up to 80% of these may be parasitized by tachinid flies (Schweizerischer Bund für Naturschutz, 1997).
Major Hostplants. Species of Epilobium, especially Epilobium dodonaei (syn. Chamaenerion angustissimum & Epilobium rosmarinifolium) in Europe. At higher altitudes in the Alps it transfers to the very similar but taller Alpine willowherb Epilobium fleischeri (syn. Chamaenerion fleischeri & Chamerion fleischeri). Epilobium colchicum (syn. Chamaenerion colchicum & Chamerion colchicum) is the preferred host in the Caucasus (Shchurov, Poltavsky & Ilyina, 2013).
If found on Epilobium dodonaei, larvae are reluctant to transfer to other species of Epilobium; however, Epilobium tetragonum is accepted with relish (Jean Haxaire, pers. comm. 2013).
Minor Hostplants. Naturalized species of Oenothera and, occasionally, Galium spp.

It should be noted that in the French Alps natural hybrids between this species and Hyles hippophaes (Esper, 1789) are regularly found on Epilobium dodonaei, with many larvae being of the pink form as shown below (J.-M. Bompar, pers. comm.)). This is not surprising as, contrary to appearance, the two species are closely related. Using mtDNA techniques, Hyles vespertilio and H. hippophaes have been shown to exhibit a closer relationship than previously understood, but both taxa nevertheless remain valid as all other analyses support a clear differentiation between them (Patzold, Marabuto, Daneck, O'Neill, Kitching & Hundsdörfer, 2021).


PUPA 35--40mm. Pale reddish-brown with translucent green-brown wings; however, the green colour and translucence fade with age. Many older pupae become pale yellowish-brown. In shape, almost identical to that of Hyles euphorbiae (Linnaeus, 1758). Contained in a fine-meshed, silk cocoon amongst debris on the ground. Overwinters as a pupa.





Tachinidae: Drino vicina (Zetterstedt, 1849), Masicera sphingivora (Robineau-Desvoidy, 1830), Nemoraea pellucida (Meigen, 1824).

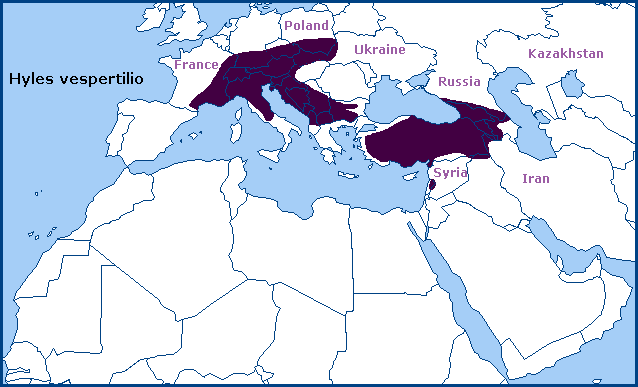
Southern and eastern France (Herbulot, 1971; Rougeot & Viette, 1978), southern Germany (Forster & Wohlfahrt, 1960; Nowotne & Köhler, 2021), Switzerland (Schweizerischer Bund für Naturschutz, 1997), Austria, northern and central Italy (Marini & Trentini, 1986; Storace, 1993; Dapporto, Fiorini, Fiumi & Flamigni, 2005; Sala & Bettini, 2005; Parenzan & Porcelli, 2006; Glerean, Deutsch, Morandini, Morin & Huemer, 2022; Babunco, iNaturalist 2024), Czechia (Šuhaj & Hudeček, 1997), Slovakia, eastern Poland, western Ukraine (Danner, Eitschberger & Surholt, 1998), western Hungary (Sáfián & Hadarics, 2005; Andras Ambrus, iNaturalist 2018), western Yugoslavia/Slovenia/Croatia/Bosnia and Herzegovina/Serbia (Andus, 1986; Koren et al., 2022; Koren & Šašić, 2023), Montenegro (Koren et al., 2022), Albania (Rebel & Zerny, 1932; Koren et al., 2022; Beshkov, Nahirnić-Beshkova & Jakšić, 2024), the Republic of North Macedonia (Huemer et al., 2011; Krpač et al., 2019; Koren et al., 2022), western and southern Bulgaria (Ganev, 1984; Pittaway, pers. obs. 2009; Hristova & Beshkov, 2016; Koren et al., 2022), and northern Greece. Also, western Turkey (Koçak & Kemal, 2018) eastward to Transcaucasia (Milyanovskii, 1959; de Freina, 2012; Zagorinsky, Gorbunov & Sidorov, 2012; Shchurov, Poltavsky & Ilyina, 2013; Didmanidze, Petrov & Zolotuhin, 2013; Streltzov et al., 2022) and Daghestan (Ilyina & Poltavsky, 2018; Oleg Kosterin, iNaturalist 2022), including the Republic of Georgia (Didmanidze, Petrov & Zolotuhin, 2013; Streltzov et al., 2022; Giorgi Iankoshvili, iNaturalist 2024), Armenia (Didmanidze, Petrov & Zolotuhin, 2013) and Azerbaijan (Didmanidze, Petrov & Zolotuhin, 2013; Snegovaya & Petrov, 2021). A small disjunct population also occurs in the mountains of Lebanon (Pittaway, 1993).
In 1996 this species was reported from the central French Pyrenees from the vicinity of Lake Gloriettes (Jean-Joseph Démotier/Jean Haxaire, pers. comm. 2013). However, an extensive search by Jean Haxaire in 2015 failed to find Hyles vespertilio in this area, which was also devoid of Epilobium. This situation requires further investigation.
Recorded with certainty only once from Romania, from Tusnad (König, 2003).
Danner, Eitschberger & Surholt (1998) indicated that this species could be extinct in Germany, but an adult was caught at light on 26 August 2017 west of Riedlingen in Baden-Württemberg (leg. Rudolf Schick), and several have been captured since then (Nowotne & Köhler, 2021).
On 4 September 2003 an adult was recorded from Scalloway on the Shetland Islands, Scotland, during a period of southeasterly winds (Walterson, 2003). A perfect adult of Daphnis nerii was also recorded from these islands ten days later after further southerly winds, along with several other migrant moths.
In the North Caucasus, Russia, this already rare species has become threatened due to the nature of its biology, i.e. the caterpillars develop on fireweed (Epilobium) plants growing on shingle bars in the beds of mountain rivers which flood regularly. Populations in the western Caucasus are more at risk than those in the eastern Caucasus (Daghestan) due to the intensive use of riverbeds (Shchurov, Poltavsky & Ilyina, 2013).
Extra-limital range. None. Endemic to the western Palaearctic region.
 Return to species list
Return to species list