| Genus | Western Palaearctic Species/subspecies | Eastern Palaearctic Species/subspecies | Nearctic Species/subspecies |
|---|---|---|---|
| Sphinx | ligustri ligustri | ligustri ligustri | drupiferarum |
| Smerinthus | ocellatus | planus | cerisyi |
| Hemaris | fuciformis | affinis | thysbe |
| Hyles | gallii | gallii | gallii |
This region has four distinct seasons -- spring, summer, autumn and winter, the last of which is relatively long and cold, with severe frosts. These climatic characteristics are not confined to the Holarctic as they can also be found in the southern temperate areas of the Australasian and Neotropical regions (the Holantarctic region of Takhtajan (1986)); however, the fauna and flora of these latter regions differ markedly from that of the Holarctic for historical reasons.
Zoogeographical regions often have definable boundaries due to physical barriers, such as mountains, deserts or water. The Palaearctic is now separated from the Nearctic by the Atlantic Ocean on one side and the Pacific Ocean and Bering Straits on the other. However, where no such barriers exist, each region gradually merges with the next, pockets of one extending some way into the other due to variable environmental conditions. Such transitional or eremic zones may themselves have certain definable characteristics and are often classified as distinct regions. The desert between the Palaearctic and Afrotropical regions is one such zone, and is known as the Afroeremic zone (de Lattin, 1967) or, more correctly, as the Saharo-Arabian subregion and Takhtajan (1986) for more details).
A zoogeographic breakdown of the present-day sphingid fauna of the western Palaearctic provides interesting information.
CLIMATIC FACTORS AFFECTING DISTRIBUTION
Apart from geographical barriers, climate is the other major influence that determines these regions and, consequently, hawkmoth distribution. The main climatic factors are maximum and minimum average temperatures; hours and strength of sunshine; rainfall (amount and periodicity); humidity; length of seasons; severity of frosts; evaporation rates; and wind (Krebs, 1972). A species' spatio-temporal variation will match climatic conditions by means of specific morphological, behavioural and physiological adaptations (Hengeveld, 1990). Competition, geographical barriers and historical factors will determine if a potential climatic range can be fully utilized. However, historical explanations should always be considered when a distribution pattern cannot be fully explained in terms of responses to contemporary environmental conditions. A species could, in theory, occur in a certain region, but may not yet have been able to reach it (Krebs, 1972). Limited dispersal capacity can cause a species to be absent even when conditions are optimal for it (Hengeveld, 1990). It should be remembered, however, that climatic barriers act in two ways: directly on the hawkmoths' ability to tolerate certain climatic conditions; and indirectly, by acting on the hostplant.
Temperature is probably the main climatic factor affecting distribution (Krebs, 1972; Lawing & Polly, 2011), as can be seen by correlating January and July isotherms with the distribution of individual species; however, laboratory experiments are needed to establish if such deductions are correct, although Hengeveld (1990) and Krebs (1972) provide conclusive evidence for species other than sphingids. Agrius convolvuli appears unable to establish itself in areas where the January mean temperature falls below +4°C, although it is quite capable of breeding over most of Europe during the summer months. Sphinx ligustri appears to have the reverse problem in that it cannot survive in areas where the July and January temperature means exceed +30°C and +4°C, respectively. Marumba quercus appears to be limited both by temperature and hostplant distribution. Its southern distribution is confined to mountain chains where oak (Quercus) grows. Its northern limit correlates very well with the +20°C July isotherm; however, its eastern limit in the Ukraine and Russia appears to be limited by the -12°C January isotherm.
As Hengeveld (1990) points out, although extreme conditions are important in limiting a species' range, these should not be estimated by their short-term deviations from mean values. Just because a species' range margin appears to coincide with a given isotherm, this does not mean it cannot tolerate higher or lower temperatures for short periods. Even though some species can remain unaffected by large short-term deviations, differences between average conditions across range boundaries often involve only 1°C or 2°C or less.
Unlike some moth species which are found worldwide, no hawkmoth is cosmopolitan although some genera such as Sphinx are very widely distributed (Eurasia, North America and South America). Some have large, continuous ranges, e.g. Laothoe amurensis, which occurs right across the Palaearctic region from Poland to Japan. If such a range is interrupted in places, as in the case of Sphinx ligustri, which is found in Europe, Asia Minor and Central Asia, and then eastern Asia with no population in-between, it is termed disjunct. Some sphingids are Holarctic, occurring right across the temperate regions of both Eurasia and North America, e.g. Hyles gallii. A species which is confined to a specific area and is found nowhere else is termed endemic: Hyles vespertilio is endemic in the western Palaearctic; Hyles dahlii in Corsica, Sardinia and the Balearic Islands. Where a small population of one species has been left isolated when the parent population retreated in the face of changing climatic conditions, it is known as a relict population. Hyles euphorbiae has one such in the mountains of south-west Arabia (Pittaway, 1987; Meerman, 1988b)) and Sphingonaepiopsis gorgoniades another in what was Yugoslavia (von Mentzer, 1974).
Because a species has a certain range today does not mean that this has always been so nor that it will remain so. However, apart from relict populations, subspecies and generic relationships, there is little direct evidence to support this view. Nevertheless, the dependence of specific sphingid species on certain hostplants, ecosystems and biomes is recognized (Hengeveld, 1990) and the study of plant remains (particularly pollen) can provide a great deal of indirect evidence. The last quarter of a million years has demonstrated just how quickly climates can change (Zeuner, 1946; Polunin & Walters, 1985; Lawing & Polly, 2011). Biogeographical regions have shifted north, south, east and even west in response to global warming and cooling trends. In response, the ranges of both hawkmoths and hostplants must have expanded and contracted; this has been well documented for other taxa (Hengeveld, 1990). In many instances isolated populations of a species must have been left behind, cut off from the main distribution by intervening, unfavourable conditions. Many of these would have been reabsorbed when the climate improved and the parent population expanded its range to recolonize former territory. Others would have died out as environmental conditions worsened; some would have persisted.
During part of the Tertiary epoch (65--2 million years BP), Europe alternated between a typical Indo-Malay climate (wet, warm and humid) and warm, temperate conditions, characterized by the presence of Magnolia species (Polunin & Walters, 1985). There then followed a period of progressive cooling with distinct changes in vegetation. Many plant species characteristic of present-day conditions appeared, e.g. Picea, Carpinus, as well as others which today are confined to the mountains of western China, eastern Xizang Province/Tibet and Japan, e.g. Pterocarya, Liriodendron and Tsuga. Some became extinct due to further cooling, or retreated to refuge areas in the Tian Shan, Turkey and Iran, e.g. Pterocarya. Further changes produced a more contemporary European flora of Quercus, Pinus, Picea and Abies intermingled with Carya, Juglans, Pterocarya and Magnolia. By the end of the Tertiary epoch most eastern Asiatic species had vanished from Europe (c. 1 million years BP) and few plants were present which do not exist in the western Palaearctic today. During the Quaternary epoch there were at least four major and several minor ice-ages interrupted by warm interglacial periods. In the warmest periods, forests of Quercus, Ulmus, Fraxinus and Corylus formed. Wetter interludes favoured the spread of Alnus, whereas drier conditions favoured Pinus. Conversely, the glacial phases produced a decidedly tundra-like community of dwarf Salix and Betula and numerous herbaceous plants. During these periods the former forests clung precariously to the Mediterranean shores and the mountains of Central Asia, Iran and North Africa. The penultimate major glaciation commenced 150,000 years BP and produced a very distinctive tundra biome in Europe dominated by Dryas octopetala. This was followed by a short interglacial and another ice-age, the Weichselian, which commenced 110,000 years BP. This finally ended approximately 10,000 BP, to be followed by a series of well-defined warming stages, characterized by various floral regimes (Polunin & Walters, 1985).
- Pre-Boreal: 10,000 -- 9500 BP
- Boreal: 9500 -- 7500 BP
- Atlantic: 7500 -- 5000 BP
- Sub-Boreal: 5000 -- 2500 BP
- Sub-Atlantic: 2500 BP -- today
With such drastic changes occurring in the biome regimes of Europe, it is safe to assume that sphingid ranges were also profoundly affected. Even to this day, two essentially east Asian butterfly taxa, Vanessa indica (Herbst) and Apatura metis Freyer, still have relict populations in the western Palaearctic (Higgins & Riley, 1980). Did a number of eastern Palaearctic hawkmoths once occur in Europe, only to be exterminated by successive ice-ages? Are the Central Asian mountains and deserts preventing them from recolonizing Europe? After all, many of their hostplants are present as are suitable climatic conditions: the Oriental Marumba gaschkewitschii (Bremer & Grey), Marumba maacki (Bremer) and Marumba jankowskii (Oberthuer) would probably feel quite at home in Europe today, if they could get there.
The effects on sphingids of changing climates and biomes can be seen in the following documented examples. Twenty thousand years ago much of the Arabian Peninsula had a Mediterranean flora and fauna. As the climate in Europe ameliorated at the end of the last ice-age, Arabia became arid. The wildlife either died out, retreated northwards or took refuge in cooler mountainous areas (Pittaway, 1987; Larsen, 1987). Today, Hyles euphorbiae conspicua ranges from Turkey to Palestine and Iraq. Two thousand kilometres to the south a small, relict population of the same or a closely related subspecies persists in the cool, Mediterranean-like Asir Mountains of south-western Saudi Arabia. Along the border with Yemen, it has come into contact with another relict sphingid population, that of Hyles tithymali himyarensis.
Likewise, Hyles hippophaes occurs today in three isolated populations -- one in western Europe, one in eastern Europe and the Aegean and one stretching from Anatolia eastward to Mongolia (Chu & Wang, 1980a; Pittaway, 1982a). This species, as a larva, feeds on Hippophae and Elaeagnus, both shrubs in the Elaeagnaceae (Pittaway, 1993). The Palaearctic Hippophae rhamnoides is a small, drought-resistant bush adapted to poor, stony, unstable soils. It has its main distribution from Anatolia eastward to China. Here it often forms pure stands, covering whole mountain-sides and mountain river-banks. In Europe, although isolated patches of this shrub exist on coastal sand-dunes, its main distribution covers the riverine gravel-beds of southern Germany, Switzerland and south-eastern France, as well as sand-bars and gravel banks along mountain river-valleys of the Pyrenees, French Alps and northern Italy. Further east it is found in similar places in Romania. This shrub does not occur naturally in the Aegean, nor western Anatolia, but both these areas have been extensively planted in recent years with the ornamental Elaeagnus angustifolia from central and eastern Anatolia.
At the beginning of the interglacial periods, including the present one, retreating ice and scouring melt-water would have produced large areas favourable to Hippophae rhamnoides in Europe. For example, in the Holsteinian Interglacial (c. 250,000 years BP), this shrub was initially one of the dominant woody plants, even in Britain (Polunin & Walters, 1985). With improving soil conditions and a wetter climate other trees and shrubs succeeded this species, forcing it into refugia.
Hyles hippophaes may once have had a continuous distribution from Britain to China. This fragmented into two populations with the break-up of the distribution of its hostplants, giving rise to the European Hyles hippophaes hippophaes and Asian Hyles hippophaes bienerti. After the last ice-age the original European population must have not only contracted in range but also split into a western and an eastern population which, as yet, have had insufficient time to diverge morphologically or genetically. However, that this sphingid can thrive in areas from which it was previously excluded due to a lack of hostplants has been demonstrated by its recent colonization of the Aegean and western Anatolia, where subsp. hippophaes can now be commonly found on Elaeagnus angustifolia (Pittaway, 1982a). Hippophae rhamnoides ranges as far north as Scandinavia and Britain, but these regions are now too cold and wet for Hyles hippophaes. Similar mechanisms must also have acted on Deilephila porcellus to produce 'subsp.' suellus and 'subsp.' porcellus, only to reunite them in the recent past.
Small, isolated European populations also occur of such essentially Irano-Turanian species as Rethera komarovi and Sphingonaepiopsis gorgoniades. Even more extreme is the present-day distribution of Sphinx ligustri. Having probably originally spread to Europe from eastern Asia during one of the last interglacial periods, it does not now occur between the Altai and north-eastern China due to inhospitable ecological conditions. The two populations have diverged a little to produce two recognizable races (labelled 'subsp.' amurensis and 'subsp.' ligustri). Hyloicus pinastri was affected in a very similar manner. It is more than likely that 'subsp.' maurorum evolved over a period in isolation in North Africa and the Iberian Peninsula during the last ice age and that it is only since the end of that period that it and 'subsp.' pinastri, which appears to have taken refuge in south-eastern Europe during the ice age, have reunited in southern France to produce many intermediate forms. Indeed, recent studies have shown that, unlike in Sphinx ligustri, they have been apart long enough to produce two sibling species.
Isolated islands which are cut off from the mainland can also lead to speciation, but this very much depends on the mobility of a species and the extent of the marine barrier. On Sardinia, Corsica and the Balearic Islands, the Saharo-Arabian Hyles tithymali is replaced by Hyles dahlii. Both these species are derived from a common ancestor, as indicated by Meerman (1993), but as to when the initial island colonization took place and why the Pontomediterranean Hyles euphorbiae has subsequently been unable to colonize these islands remain a mystery. In the analogous butterflies Papilio saharae Oberthuer and Papilio hospiton Guenée (Pittaway et al., 1994), Papilio machaon Linnaeus has managed to establish itself on Corsica and Sardinia and both it and the endemic Papilio hospiton co-exist with little interbreeding (Pierron, 1990). However, 'islands' need not necessarily be marine; small mountain chains can also act as 'islands' when the surrounding lowlands become too hot and/or dry. The Tian Shan appear to have undergone this phenomenon several times, resulting in the formation of several endemic species and subspecies of sphingid.
With time, differential selection pressures may also produce two species in what could be termed a longitudinal cline (Hengeveld, 1990). Gene flow between a bivoltine southern race and a univoltine northern population may be sufficiently reduced to initiate subspeciation as each population adapts to its ecological niche, a feature much discussed by Krebs (1972).
It should be remembered, however, that complete speciation may not be achieved during the course of any one of the above isolation mechanisms if the selection pressures on each isolated population remain the same. Reunification could eventually either produce a merging hybrid population until fully intermixed, such as in Deilephila porcellus/suellus, or lead to complete speciation if the resulting hybrids were at a disadvantage. Selection pressures would force the two subspecies apart. During this process both populations would tend to remain allopatric and could be regarded as 'sibling species'. Such are Hyles euphorbiae and Hyles tithymali -- the evidence indicates that they are good species separated by ecological adaptations.
FLORISTIC FACTORS AFFECTING DISTRIBUTION
TYPES OF BIOMES
Like all organisms, the Sphingidae are highly specialized, with each species adapted to live in a certain type of habitat or ecosystem. The suitability of each ecosystem will depend on the presence of suitable hostplants and such critical features as altitude, rainfall, sunshine and temperature (Odum, 1971; Krebs, 1972). A number of similar ecosystems may share the same area and form a community. A collection of these is termed a 'biome', characterized by a particular 'climax community' that remains relatively stable in the typical climatic conditions of a given region. Worldwide there are many types of biome but in the western Palaearctic one finds only Tundra, Boreal Forest, Deciduous Temperate Forest, Mediterranean, Steppe and Desert (Polunin & Walters, 1985). It should be stressed, however, that biomes are rarely uniform, but comprise a complex of communities within a given range of conditions; they are the result of an equilibrium established between the organisms of a region and its climate (Hengeveld, 1990). Local differences in altitude and precipitation may also produce pockets of one biome within another. Additionally, certain ecosystems, for example riverine systems, may be found in several biomes. These often allow some hawkmoths to penetrate biomes seemingly hostile to them, such as the deserts of Central Asia and North Africa.
Map 1: Map of the principal world vegetation types.
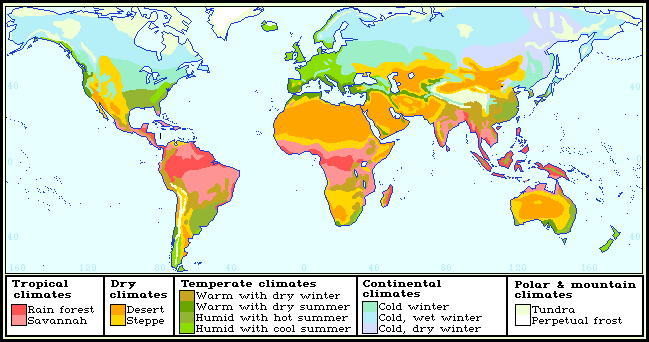
Biomes should not be confused with biogeographical regions and subregions. Although a floral region, such as the arid Irano-Turanian of Takhtajan (1986) may be dominated by steppe, several other biomes are present in varying degrees. This presents a number of problems with regard to classifying the zoogeographical affiliation of species. The mix of environmental conditions necessary for a species' survival may be present in more than one biome. Thus ranges should be viewed as broad-scale response surfaces, with the highest spatial and temporal abundances at their centres indicating those environmental conditions most favourable to the species (and hence zoogeographical affiliation). The gradual decrease in abundance towards range margins is due to a gradual progression towards more unsuitable conditions (Hengeveld, 1990). The abundance patterns of species over their ranges will manifest themselves in terms of responses to spatially varying ecological conditions. These patterns are to be expected and vary with time (Hengeveld, 1990).
TUNDRA (ARCTIC)
This region lies between the northern tree-line and permanent ice, where the average summer temperature never exceeds 10°C. For two months there is continuous daylight with little or no frost, although the subsoil remains permanently frozen (permafrost). In winter everything freezes for months on end, there is permanent snow-cover and darkness for two months. Annual rainfall averages 250mm. Winds can be very strong.
The vegetation of this region is very distinctive, being composed of dwarf evergreen members of the Ericaceae (heath family) and of Carex (sedges), Juncus (rushes), mosses and lichens. No western Palaearctic Sphingidae are adapted to this zone although some, like Agrius convolvuli, Hyles euphorbiae, Hyles gallii and Hyles livornica, migrate into it.
BOREAL FOREST (TAIGA)
This region forms an almost continuous band, some 1500km wide, right across the northern hemisphere (the Holarctic). To the north it merges with the tundra, to the south with mixed temperate forests. Its climate is profoundly influenced by cold polar air masses; only at its western and eastern extremities do moisture-laden sea winds have any ameliorating effect. Winters are cold to very cold, with average temperatures less than -7°C. Snow-cover can last up to six months during the short days of winter. In contrast, the short summers are relatively warm, with average temperatures reaching 15°C during the long days. Rainfall rarely exceeds 500mm. Thus the growing period for plants varies between 100 and 160 days.
The vegetation is dominated by conifers (Picea, Abies and Pinus, with some Larix). Often a single species of tree covers huge areas, Picea abies (Norway spruce) being the most important. In drier areas it cedes its dominance to Pinus sylvestris (Scots pine). In clearings and along rivers a number of broad-leaved trees occur, notably Betula pubescens (downy birch), Sorbus aucuparia (mountain ash), Alnus incana (grey alder) and Populus tremula (aspen). Beneath all of these there is a sparse shrub layer of various species of Salix (sallows) and members of the Ericaceae (heath family).
In the western Palaearctic two species, Laothoe amurensis and Smerinthus caecus, are adapted specifically to this zone, penetrating south only a short way into mixed temperate forests. In the same way, a number of deciduous temperate forest species (Sphinx ligustri, Hyloicus pinastri, Smerinthus ocellatus, Laothoe populi, Hemaris fuciformis, Hemaris tityus, Hyles gallii and Deilephila elpenor) occur in the mixed forest zone and may even reach the boundaries of the true Taiga forests.
DECIDUOUS TEMPERATE FOREST
Farther south, especially where humid onshore sea-winds ameliorate the more severe continental climates, monotonous Boreal forest gradually gives way to species-richer deciduous forest. In the Palaearctic, this biome is unfortunately rather limited and irregular due to the effects of several Pleistocene glaciations and human deforestation. It reaches its greatest development in central Europe, the Caucasus/Alborz, and eastern Asia (Korea, Japan and northern China), with a relict area in the western Himalaya and Tian Shan (Shchetkin, 1956). In Europe two distinct sub-biomes are recognized -- the Atlantic climatic zone and the central European climatic zone. None of the Sphingidae is specifically adapted to the former; indeed, many continental species, such as Proserpinus proserpina, Hyles euphorbiae and Hyles gallii, seem unable to tolerate the cool, cloudy and wet conditions found there. It is one of the cloudiest regions of the world with, for instance, only 1700 hours sunshine per annum in London. The average temperature difference between winter and summer is only 8--14°C: the July and January (in brackets) averages for London and Santiago de Compostella, northern Spain, are 16°C (3.8°C) and 18°C (7.3°C), respectively. Rainfall occurs throughout the year and ranges from 580mm in Paris to 165lmm in Santiago de Compostella.
Farther east, the high-pressure system of eastern Europe dominates the weather. It is less variable, sunnier, less humid and more extreme. January mean temperatures vary between -3°C (Warsaw) and -1.7°C (Belgrade). Snow-cover may last up to three months, with continuous sub-zero temperatures. By contrast, the average mid-summer temperatures can be relatively hot -- Warsaw 18.5°C and Belgrade 22.2°C. Thus the average temperature-difference between winter and summer usually exceeds 19°C. Rainfall rarely exceeds 635mm, but increases to 3000mm on some mountains. There is no distinct rainy season, rainfall being spread evenly throughout the year.
As the description suggests, the vegetation is dominated by deciduous broad-leaved trees, although certain Pinus species can form extensive mixed forests in drier areas. The woodlands are much more complex than those of the Taiga, with well-developed strata of herbaceous plants, shrubs, small trees and tall trees. The diversity of species is also much higher, resulting in a rich and abundant lepidopterous fauna. The dominant trees are various species of Quercus (oak), Fagus (beech), Tilia (lime\linden), Acer (maple), Ulmus (elm), Castanea (sweet chestnut), Fraxinus (ash) and Carpinus (hornbeam). Having escaped the repeated glaciations which decimated the European forests, those of the Caucasus and Alborz are also rich in genera formerly found in Europe, such as Zelkova (Ulmaceae), Pterocarya (Juglandaceae) and Liquidamber (Hamamelidaceae).
The greatest number of western Palaearctic sphingid species inhabit this biome. Besides those mentioned under Boreal forest above, others are Mimas tiliae and Deilephila porcellus. Some Mediterranean and steppe species (Marumba quercus, Proserpinus proserpina, Hyles euphorbiae and Hyles vespertilio) penetrate this region from the south. They tend to be found in the driest, sunniest and warmest locations, in other words, in localities where the microclimate is similar to that of their centres of origin, a feature also noted by Hengeveld (1990) for other taxa. Nearly all colonies of these species are confined to very warm south-facing banks and hillsides at low elevations in central Europe, where the larvae can bask in hot sunshine. Large caterpillars can so increase their body temperature up to 10°C or more above that of their surroundings (Reavey & Gaston, 1991), and the level of opportunity for basking can probably make or break the survival of a colony and probably limits the spread of such species as Hemaris croatica. Conversely, most temperate-forest species can also occur farther south in favourable situations, such as along river valleys and mountain chains.
MEDITERRANEAN
This is the most distinctive of those found in the western Palaearctic. The winters are wet and mild with an average temperature of 6°C. Summers are hot and sunny (2500 hours sunlight per annum), with temperatures averaging 21°C. Most of the rain falls as short storms between September and April, and mid-summer is marked by two or three rainless months. The amount of precipitation varies markedly and depends on proximity to the coast, mountain ranges and prevailing winds. Skopje in the Republic of North Macedonia receives 480mm per annum; just across the mountain ridge, on the southern coast of Montenegro, rainfall can exceed 4000mm.
The vegetation is dominated by sclerophyllous trees and shrubs with hard, evergreen leaves. Herbaceous plants which die back during the summer months and a rich diversity of spring and autumn annuals are also characteristic features.
A number of sphingids are resident in this biome, with many penetrating from here northwards into warmer regions of deciduous forest. Those characteristic of the Mediterranean region are Hemaris dentata, Hemaris croatica and Hyles nicaea. Others, such as Akbesia davidi, Smerinthus kindermannii, Clarina kotschyi and Rethera brandti, are of Irano-Turanian origin and are confined to the hotter and less humid eastern Mediterranean subregion and penetrate only a short distance westwards. Although they all occur within the Mediterranean biome, each of these species inhabits a distinct ecosystem. However, there is a large overlap in the ranges of Irano-Turanian and Mediterranean species throughout the Middle East and southern Central Asia due to an intermingling of biomes brought about by variations in rainfall, aspect, altitude and the presence of rivers.
A few cold-tolerant tropical Sphingidae have also become established along the southern periphery of this region and others migrate into it. These include Agrius convolvuli, Acherontia atropos, Acherontia styx, Daphnis nerii, Sphingonaepiopsis nana, Hippotion osiris, Hippotion celerio and Theretra alecto. The survival of some of these, such as Daphnis nerii and Hippotion celerio, would be very unlikely were it not for continuous migrations from the tropics, many of which penetrate far to the north.
STEPPE (PRAIRIE)
This is a temperate zone biome, also referred to as 'Pannonic Steppe' in central Europe or, in South America, as 'Pampas'. Rainfall is low (400--500mm.) and insufficient to support trees away from sources of groundwater. Most of the rain falls between May and June (earlier in Anatolia), with July and August experiencing drought conditions. There is a large winter/summer temperature range of more than 19°C, the winters being cold and dry. For example, in Bucharest the average July and January temperatures are, respectively, 22.8°C and -3.9°C.
The vegetation is dominated by grasses interspersed with various species of Fabaceae and Asteraceae. Throughout this region, pockets of Mediterranean or temperate deciduous trees may also occur where there is sufficient water. In some areas, sparse Quercus (oak) woodland can dominate.
In Europe, Pannonic Steppe is found in central Hungary and southern Romania, and across the southern part of Russia, Moldova and the Ukraine bordering the Black and Caspian Seas. In the Middle East, much of the Anatolian and Iranian plateaux, northern Syria, Lebanon and Iraq have large areas of steppe intermixed with Mediterranean scrub (Pontic Steppe). Similar conditions also exist along the southern foothills of the Atlas Mountains in North Africa.
A number of Irano-Turanian sphingid species are very characteristic of this biome and often occur as isolated refuge populations. These remained when the once predominant steppe habitat retreated eastward as the climate grew wetter after the last ice-age. Among such are Rethera komarovi, Sphingonaepiopsis gorgoniades, Hyles centralasiae, Hyles zygophylli and Hyles hippophaes. Hemaris croatica and Hyles nicaea frequent both Pontic Steppe areas and Mediterranean regions.
DESERTS
Across the whole of the southern part of the western Palaearctic, the Mediterranean and Steppe biomes gradually merge into deserts (Sbordoni & Forestiero, 1985) to form the Afroeremic or Saharo-Arabian subregion (Takhtajan, 1986; Larsen, 1987) in North Africa and Arabia, and the southern part of the Irano-Turanian subregion in Asia. Rainfall here is erratic, rarely exceeds 200mm per annum, and usually falls in the winter. The summers are usually rainless and often cloudless, with average July temperatures exceeding 30°C. The winds are strong and the evaporation rates so high that the unstable soils tend to be badly degraded.
Most plants are annuals which, after a downpour, grow rapidly, flower and set seed. Perennial plants tend to be xerophytic and widely spaced with extensive root systems.
The most widespread true desert sphingid is Hyles livornica. Its main breeding cycle coincides with the winter and spring flush of annual flowers; its larva is polyphagous and the adult moth highly migratory -- all adaptations to a desert life. The one other true desert species is a sedentary Saharo-Arabian endemic, Hyles tithymali deserticola, which has managed to adapt to the Saharan and Arabian deserts, where its larva feeds on xerophytic species of Euphorbia.
MOUNTAINS
These are not, strictly speaking, true biomes, but mountain ranges do have a considerable effect on the climate, vegetation and fauna of the region. There tends to be a rapid succession of different species as the zones of vegetation change with altitude. Each of these zones often corresponds with a similar one found with increasing latitude. Thus in the Alps one can find both Boreal and Tundra biomes in what would otherwise be classified a temperate deciduous forest region.
Such a phenomenon has a marked effect on the Sphingidae of this region. Mimas tiliae does not occur above 1500m in the Alps, but in Iran it is rarely found below this altitude. Indeed, many species and subspecies in the southern part of the western Palaearctic are confined to mountain chains. It is very unlikely that Hemaris fuciformis or Hemaris aksana would have survived the post ice-age drying-out and desiccation of North Africa were it not for the Atlas Mountains. Such has been the importance of the Tian Shan, Pamir and Hindu Kush in Central Asia as long-term refugia, that several distinct species and subspecies of Sphingidae have evolved in isolation there, e.g. Dolbina grisea, Laothoe philerema, Hemaris rubra, Hemaris ducalis, Acosmeryx naga hissarica, Rethera afghanistana, Sphingonaepiopsis kuldjaensis, Hyles nervosa, Hyles salangensis and Hyles chamyla.
OTHER FACTORS AFFECTING DISTRIBUTION
Four main factors may determine a contraction in range: a change of climate; human activity; a change of genetic constitution; and increased competition (Krebs, 1972). In recent times, the second factor has proved a major force, as has been discussed by Hengeveld (1990).
In the 1960s, when the author was cycling around country lanes in south-east Austria during August, several fully grown larvae of Hemaris tityus and Deilephila porcellus could be seen and picked up on the road every day as they went in search of pupation sites. At that time the roadside vegetation was cut twice each summer. Additionally, overgrown boundary strips between the fields yielded countless larvae of Macroglossum stellatarum, Deilephila elpenor and Deilephila porcellus at night, and numerous adults at garden flowers and lights. By the 1980s the roadside verges were being cut every four weeks and the boundary strips had vanished, and so had most of the hawkmoths -- both larvae and adults. In this area no increase in the use of insecticides was noted and the damage appeared to be purely mechanical (Pittaway, 1993). Similarly, flood-control measures in the Swiss and French alpine valleys have reduced the amount of Hippophaes rhamnoides and with it the numbers of Hyles hippophaes. In most cases it is the physical destruction of habitats which is more important than the much discussed use of pesticides. This is especially so at the periphery of a hawkmoth's range, its 'range margin'. Over the last 40 years Hemaris tityus has suffered a dramatic drop in numbers in Britain (Gilchrist, 1979; Waring, 1992) and is all but extinct in Holland (Meerman, 1987). This species favours as breeding sites those lowland meadows near rivers which are now regarded as prime agricultural or building land. The same fate appears to have befallen relict populations of Hemaris croatica in central Europe and european Russia.
The importance of range margins has been demonstrated by Hengeveld (1990). They can be interpreted in terms of the sum of several variables which vary independently in space, replacing and/or counterbalancing each other at one or more locations. However, these variables should be sampled across the complete range of a species, and encompass areas of both abundance and scarcity if one is to obtain a true picture of the factors limiting a species' range.
An expansion in range can also occur due to human activity, either directly by the introduction of a species into a new area, or indirectly by the alteration of local climatic conditions or the availability of hostplants (Krebs, 1972). Within the last ten years, Hyles hippophaes has colonized the Aegean from (?)Romania in response to widespread planting of the amenity shrub Elaeagnus angustifolia (Pittaway, 1982a). The popularity of species of Tilia (limes), Ulmus (elms) and Populus (poplars) as ornamental trees in towns and cities has, no doubt, benefited both Mimas tiliae and Laothoe populi. Both are very common in city suburbs and rarer in rural areas. Within recent times Acherontia styx has spread right across Saudi Arabia. Originally confined to the Arabian Gulf oases, with the development of Saudi Arabia and the spread of gardens and farms, it adopted various garden shrubs as larval hostplants, appearing first in 1979 in Riyadh and then in 1982 in Jeddah (Wiltshire, 1986; Walker & Pittaway, 1987). How long before it crosses the Red Sea and enters Africa? Even the normally sedentary Proserpinus proserpina appears to be expanding its range in Europe in response to some, as yet unknown, change in its biology or ecology. During the last decade, the species has colonized eastern Belgium and individuals have been captured in Britain (Pratt, 1985) and Corsica (Guyat, 1990).
Although much has been said about the expansion and contraction of a species' range in response to changing environmental conditions, some species have managed to survive intermittent inhospitable regimes and recolonize much of their former range thereafter. But where did the refugees survive? De Lattin (1956; 1967) has reviewed and analyzed this question in detail.
THE ORIGINS OF THE WESTERN PALAEARCTIC SPHINGIDAE
Duing the last ice age that biome richest in hawkmoth species, the temperate deciduous forest, clung precariously to the Mediterranean and Black Sea shores, and the Caucasus and Tian Shan, and so, presumably, did many Sphingidae. These are known as 'refuge' areas. Each had its own characteristic fauna which developed in isolation. With a gradual increase in temperature many species spread out from these land-locked islands to colonize their territories again. This type of expansion is clearly evident in Scandinavia where no hawkmoths survived the glaciations. Since then, the Manchurian refugium species Laothoe amurensis and the Pontomediterranean refugium species Laothoe populi have recolonized that area. This has been due to a breakdown in barriers between the refugia. Of course, during the periods of isolation speciation may or may not have occurred if one species had been trapped in two or more refugia, i.e. it was polycentric. The area encompassing the Tian Shan and Pamir Mountains (the Turkestan centre of de Lattin (1967)) gave rise to several new species and subspecies, such as Laothoe philerema, Sphingonaepiopsis kuldjaensis, Hyles chamyla and Acosmeryx naga hissarica. The first three are closely related to Laothoe populi, Sphingonaepiopsis gorgoniades and Hyles hippophaes respectively. Laothoe philerema now appears unable to spread into neighbouring regions occupied by Laothoe populi or Laothoe amurensis, and these two species have been unable to penetrate the Tian Shan.
In the western Palaearctic a number of Pleistocene refugia are recognized -- the northern Mediterranean littoral (Italy, Spain); the Pontomediterranean refugium (Greece and N. Turkey); the southern Mediterranean littoral (North Africa); the Syrian refugium (N. Syria and N. Iraq); the Caspian refugium (Crimea and Caucasus); the Iranian refugium (Iran); and the fragmented Turkestan refugium (Kyrgyzstan/Tajikistan/China). Most of the region's hawkmoths survived the ice-ages in one or more of these areas; a few eremic species appear to have even survived in the Afroeremic (North African Sahara), Syroeremic (central Arabian Peninsula), Turanoeremic (Transcaspia) and Mongoloeremic (Xinjiang) refugia. Others, such as Daphnis nerii and Acherontia atropos, almost certainly became extinct and recolonized from their tropical ranges. Two, Smerinthus caecus and Laothoe amurensis, came from the very important Manchurian refugium. Several species evolved distinct (sub)species during their period of isolation, such as Hemaris aksana (from Hemaris tityus) in North Africa. The former has remained isolated (and has speciated) by retreating into the isolated Atlas Mountains. However, it should be remembered that, on occasions, the terms 'refuge population' and 'relict population' become synonymous. But what are the recent origins of most of the present western Palaearctic sphingid fauna? This can best be explained by studying their zoogeographic affiliation.
Table: The number of western Palaearctic sphingid species and subspecies by present-day zoogeographic affiliation and status.
