Type species: Sphinx atropos Linnaeus, 1758.
An Old World genus comprising three species, of which two occur in the region.

IMAGO: Large and heavily built, with relatively large dark forewing. Hindwing yellow with black submarginal lines. Dorsal surface of thorax with a skull-like marking. Proboscis shorter than thorax, stout and hairy, with a dorsal opening before tip. Labial palpi well separated. Antenna stout, of even thickness, with a fine terminal hook. Abdomen with yellow rib-like markings. Hindtibia with two pairs of spurs. Mid- and hindtarsi strongly compressed, without a ventral brush of long bristles at base. Pulvillus absent; paronychium reduced to form a short stumpy lobe. Squeaks if molested.
Genitalia. Male valva with a patch of large, multi-dentate friction scales exteriorly; sacculus short with two teeth.
OVUM: Oval, pale green or bluish, small for size of moth.
LARVA: Polymorphic, with a distinctly granulose yellow or light brown, downward-curving, S-shaped horn. Bears pronounced bi-coloured oblique lateral stripes and dorsal spots in the final instar. Clicks mandibles if molested.
PUPA: Smooth and glossy, with proboscis fused to body; transverse file-like ridges at base of proboscis. Cremaster broad, with two short apical spines.
HOSTPLANT FAMILIES: Trees, shrubs and herbaceous plants, mainly in the Solanaceae, Bignoniaceae, Verbenaceae and Oleaceae.
UK: Death's Head hawkmoth, F: Sphinx Tête-de-Mort, D: Totenkopfschwärmer, RUS: Mertvaya Golova, S: Dödskalle-Svärmare; Dödskallesvärmare, NL: Doodshoofdpijlstaart; Doodshoofdvlinder, CZ: Smrtihlav obecny; Lišaj smrtihlav, H: halálfejes lepke; halálfejes szender, E: esfinge de la muerta; calavera; cabeza de muerto, PL: Zmierzchnica trupiaglówka, FIN: Pääkallokiitäjä, I: sfinge testa di morto, HR: mrtvačka glava, DK: Dødningehoved, N: Dødninghode, EST: Tontsuru.
Sphinx atropos Linnaeus, 1758, Syst. Nat. (Edn 10) 1: 490.Type locality: Europe.
[Further details on this species, as well as photos of all stages, can be found on Lepiforum.]


Palaeotropical; Afrotropical region. Has become part of the present-day Holomediterranean faunal element.

Wingspan: 90--130mm. As illustrated. Colour somewhat variable, especially in intensity, clarity of markings and the presence of black hindwing bands. Individuals with no skull-like marking are occasionally found (f. obsoleta Tutt), as are melanistic individuals lacking all yellow colour. Very difficult to confuse with any other species except Acherontia styx (Westwood, 1847). (See under Acherontia styx for distinguishing features between the two species.)
In Europe this moth was, in the past, much feared for several reasons and hence seen as 'a harbinger of epidemic illnesses and pestilences'. It was a large, dark creature of the night with glowing eyes and bearing an outline of a skull or death-mask on the thorax. The yellow and black bands on the abdomen resembled 'ribs', which were hidden under cloak-like wings which draped around the body. If disturbed it would squeak and hop around like a demented demon, the noise being seen as an audible warning of the approach of death. In rural France, even contact with a single wing scale was reputed to cause blindness. This fear is reflected in the vernacular names given to this moth across Europe, from England to Russia (Kitching, 2006; Gandy, 2016).


The mottled forewing coloration of this species breaks up its outline when it rests by day on tree-trunks and walls or on leaves on the ground, with its wings held tent-like over the body. It is in such situations that mating pairs can sometimes be found, 'tail to tail', 'side by side' or even 'belly to belly', from late evening to the following morning, although some separate after only a few hours.
Frequents cultivated areas where potato is grown and open scrub with solanaceous plants, tending to prefer drier and sunnier locations. Not as common as it used to be, due to the use of insecticides.

Whether in search of a mate, or feeding, Acherontia atropos is active from dusk until after midnight. It is attracted to light and occasionally to the nectar of potato, tobacco and sweet-william flowers, and orange blossom. Many individuals have been seen to frequent bee-hives where, upon entry, they usually feed undisturbed on the honey, puncturing combs with their short, sharp proboscises. Moritz et al. (1991) have shown that this species makes itself 'chemically invisible' to honeybees by mimicking the cutaneous fatty acids of its hosts. According to Daniele Besomi (pers. comm. 2016) the moths get away with this behaviour most of the time, but occasionally the chemical trick does not work. The bees sometimes realize they are impostors and kill them. Then, being unable to drag them out of the hive, and not wanting them to rot in-situ, they suck them dry and embalm the hard parts with propolis. The below photo is of a specimen that was found in one of the hives of Daniele Besomi on 29 August 2016. There are two holes in the abdomen, one on the top (where it attaches to the thorax) and one on the bottom. These may have been made by the bees in order to empty them.

If disturbed while feeding, or for that matter at any other time, the adults raise their wings, run and hop around, while emitting high-pitched squeaks (Kitching, 2006). This is especially noticeable in males that have recently arrived at a light source and have not yet settled down. The source of these has been investigated by Zagorinsky, Zhantiev & Korsunovskaya (2012). Often this activity is accompanied in males by a mouldy smell given off by abdominal hairpencils (Birch, Poppy & Baker, 1990).
Migrant and multivoltine; in its resident range, normally from March/April to August/September, although occasional specimens are on the wing in February and October. Continuous-brooded from North Africa southwards, where the winter is passed as a larva or pupa. Farther north, mainly during August, September and October as the offspring of immigrants in June and July.
OVUM: Oval, 1.5 x 1.2mm, matt green or greyish blue with a slight polygonal network on its surface (evident only under magnification). Changes to golden buff just before emergence. Generally laid singly, low down underneath old leaves of the hostplant.

LARVA: Full-fed 120--130mm. Polymorphic: green, brown, yellow or pale creamy-yellow.






On hatching, the 6mm-long larva consumes its egg-shell. Initially light, frosted yellow (the frostiness being imparted by small, closely packed, pale yellow tubercles and lines), it quickly darkens to green after feeding on leaf material. At this stage the horn is black and disproportionately long, with a bifurcate tip. In the second instar, thorn-like tubercles adorn the dorsal surface of the thoracic segments. In the third instar, blue or purple edging to the yellow lateral stripes appears and the still disproportionately large horn is now yellow with a black base. After the next moult the colours become more vivid: dark purple spots cover the dorsal surface from abdominal segment 1, with yellow replacing all the black pigmentation of the horn. However, it is only in the final instar that the horn assumes its characteristic down-curved shape, the dorsal tubercles vanish and yellow and brown colour-forms appear in addition to the more normal green.



Initially, the small larva clings to a vein on the lower surface of a leaf, nibbling small holes in its side between periods resting in a sphinx-like posture. Later, size and weight necessitate the use of a whole petiole or stem as a resting platform, with the well-camouflaged larva hanging from it. Whatever their size, larvae are extremely inactive, moving only in order to find a fresh leaf. This leads to severe and noticeable denudation over a small area. If disturbed, large larvae will repeatedly click their mandibles and may even bite an attacker severely.
Prior to pupation, the fully-grown larva darkens over a period of several hours, during which stage it anoints its whole body with 'saliva'; this appears to hasten the darkening process (see also Agrius convolvuli). This completed, a suitable location for pupation is sought.

Generally found from July to October in Europe, but also throughout the winter in North Africa.
Major Hostplants. Most Solanaceae, especially Solanum tuberosum (potato), Solanum dulcamara, Solanum melongena, Lycium europaeum, Lycium barbarum, Physalis angulata, Hyoscyamus, Atropa belladonna (deadly nightshade), Nicotiana tabacum (tobacco) and Datura stramonium (thorn-apple/Jimson weed).
Minor Hostplants. Verbenaceae (Vitex agnus-castus), Oleaceae (Jasminum, Ligustrum, Olea, Schrebera alata and Fraxinus), Beta vulgaris, Buddleja, Cannabis sativa (Gladis & Alemayehu, 1995), Catalpa bignonioides, Malus pumila, Nerium oleander, Pyrus communis, Sambucus, Sesamum indicum, Stachytarpheta jamaicensis, Tecomaria capensis, Tecoma stans and many other plants. An occasional local pest of olive trees (Turati & Zanon, 1922), even to this day (Stavrakis, 1976). Also, occasionally reported from cultivated Cucurbitaceae (Garbowski, 1892).
Recorded in the Canary Islands from Cussonia, Podranea riscasoliana, Spathodea campanulata, Tabebuia, Cordia sebestena, Jasminum, Datura, Nicotiana glauca and various species of Clerodendrum (van der Heyden, 1989; van der Heyden, 1991; Retzlaff, 1995).
PUPA: 75--80mm. Rich, glossy, mahogany-brown; very active. Proboscis fused to and flush with abdomen. Pupation normally takes place 15--40cm deep in a large, smooth-sided cavity. The main overwintering stage in its resident range; rarely survives European winters.


Ichneumonidae: Amblyjoppa fuscipennis (Wesmael, 1845), Amblyjoppa proteus (Christ, 1791), Callajoppa cirrogaster (Schrank, 1781), Callajoppa exaltatoria (Panzer, 1804), Diphyus longigena (Thomson, 1888), Diphyus palliatorius (Gravenhorst, 1829), Ichneumon cerinthius Gravenhorst, 1820, Netelia vinulae (Scopoli, 1763); Tachinidae: Blepharipa pratensis (Meigen, 1824), Compsilura concinnata (Meigen, 1824), Drino (Zygobothria) atropivora (Robineau-Desvoidy, 1830), Exorista sorbillans (Wiedemann, 1830), Masicera pavoniae (Robineau-Desvoidy, 1830), Winthemia cruentata (Rondani, 1859), Winthemia rufiventris (Macquart, 1849).
The commonest larval parasitoid in Sicily is Drino atropivora [syn. Sturmia atropivora Robineau-Desvoidy, 1830].

Drino atropivora has even been recorded as a parasitoid from Malta (Schembri, Gatt & Schembri, 1991).

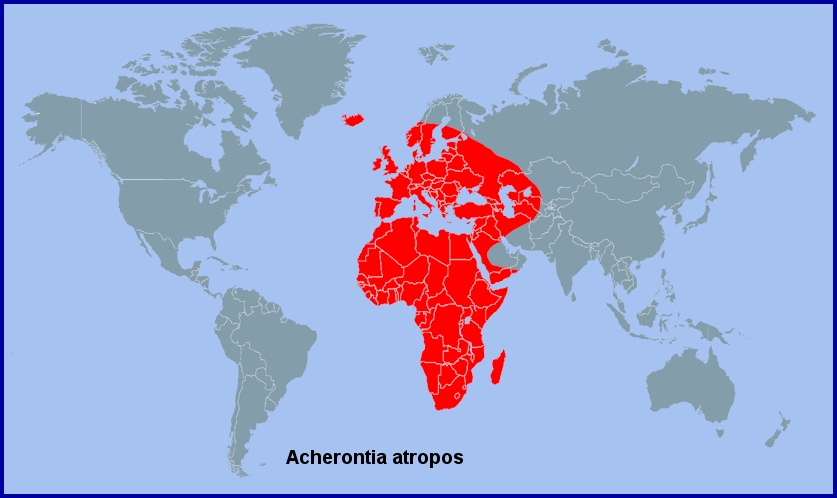
An Afrotropical species which extends north to the Mediterranean (including the whole of North Africa and the Middle East (Gentry, 1965)) and across Cyprus (Lewandowski & Fisher, 2002), Turkey (Daniel, 1932; de Freina, 1979), the Republic of Georgia (Didmanidze, Petrov & Zolotuhin, 2013), Daghestan, Russia (Didmanidze, Petrov & Zolotuhin, 2013), Azerbaijan (Snegovaya & Petrov, 2021) to north-eastern Iran (Bienert, 1870; Sutton, 1963), the Ukraine (Dubatolov, [1999]; Khalaim, 2022), Turkmenistan (Danov & Pereladov, 1985; Danner, Eitschberger & Surholt, 1998), Mesopotamia/Iraq (Wiltshire, 1957), Kuwait (Pittaway, 1993; Amr, 2021) and western Saudi Arabia (Wiltshire, 1986). It is also found in the Canary Islands, Madeira (Martin, Barnett & Emms, 2000) and the Azores (Meyer, 1991), and throughout Europe as a migrant, including Iceland (Wolff, 1971). Recorded as a vagrant as far north as Arkhangelsk Oblast (Kozlov, Kullberg & Zverev, 2014), Izvail' (Tatarinov, Sedykh & Dolgin, 2003) and Udmurtia (Bolshakov & Okulov, 2014) in European Russia, and as far east as Pavlodar in north-eastern Kazakhstan, where an example was collected in September 2006 (Dubatolov & Titov, 2011).
Although resident in parts of southern Europe, a few individuals are found each year in central and northern Europe and, in some seasons, there are marked invasions, with larvae occurring in abundance during September and October. In the exceptional years of 2022 and 2023, numerous adults and larvae were found in the UK and the Netherlands. A few occasionally survive the winter as pupae, producing adults in the spring.
Extra-limital range. The entire Afrotropical region, including the Cape Verde Islands (Traub & Bauer, 1982; Baliteau & Baliteau, 2011; Tennent & Russell, 2015), Ascension Island (Robinson & Kirke, 1990), southern Arabia (Yemen north to Salalah, Oman), and the western Indian Ocean islands of the Comoros, Madagascar, Mauritius, Réunion, the Seychelles (Fletcher, 1910; Matyot, 2005; Lawrence, 2015) and Socotra (Fletcher, 1910).
 Return to species list
Return to species list