

Sphinx oldenlandiae Fabricius, 1775, Syst. Ent.: 542. Type locality: not stated.
Synonym. Sphinx oldenlandiae Fabricius, 1775.
Synonym. Sphinx drancus Cramer, 1777.
Synonym. Deilephila argentata Stevens, 1828.
Synonym. Chaerocampa sobria Walker, 1856.
Synonym. Chaerocampa puellaris Butler, 1876.
Synonym. Deilephila proxima Austaut, 1892.
Synonym. Theretra oldenlandiae fuscata Gehlen, 1941.
Synonym. Theretra oldenlandiae olivascens Inoue, 1973.
[Further details on this species in Japan, as well as photos of many stages, can be found on Digital Moths of Japan as well as Moths of the southern Shikoku, Japan.]
Wingspan: 54--80mm. Slightly sexually dimorphic, with males being more silverish than females. The head and thorax are earth brown, with a greyish-white dorso-lateral stripe extending from the palpus to the end of thorax. There is also a broad dorsal streak of the same colour, which extends down onto the abdomen: the tegulae are fringed with silver. The greyish-brown abdomen bears a double line of silver dorsally, and an obscure ochreous lateral stripe. The greyish-brown forewings have several oblique bands running from the inner margin towards the tip. The central one is whitish-brown, edged above with silver, below with dark brown. The costal area is greyish-brown, becoming more whitish towards the base. This area is separated from the central silver edging by a dark brown band. The marginal and submarginal area are also greyish-brown. Thus similar to Theretra cajus (Cramer, 1777), but pale bands and lines less white and lacking the dark line running along Cu from the wing base to the origin of Cu1. The hindwing uppersides are blackish-brown with a paler brown submarginal band of variable shade and distinctness, sometimes vary faint.
In the male genitalia, uncus and gnathos similar to Theretra margarita (Kirby, 1877), but gnathos has an obvious medial tooth. Harpe weakly curved, spatulate in dorsal view. Phallus with band-like armature broader and longer than in Theretra margarita, teeth less prominent, right process rather short.
There are several similar species: Theretra cajus, which has a black triangle in the anal angel of the forewing; it is also confined to southern Africa from where Theretra oldenlandiae is absent. In Theretra margarita from Queensland, Australia, and Theretra silhetensis (Walker, 1856) from India and South East Asia, there is but a single silver dorsal line along the abdomen. In Theretra japonica (Orza, 1869), the forewing oblique bands are much less distinct and partly fused; it also lacks the silver dorsal lines on the abdomen. In Hippotion celerio (Linnaeus, 1758), the hindwings are pink with black venation.




This adaptable migrant species inhabits forest margins, open scrub, orchards, cultivated landscapes and suburban gardens. The moth is often caught feeding at flowers at dusk, and is also attracted by light (Bell & Scott, 1937). In the Russian Far East, a lowland species of deciduous woodland characterized by Quercus mongolica (Izerskiy, 1999b).
China: iv (Jiangxi); vi (Guangdong; Yunnan; Hong Kong; Hubei); 20.vii (Shaanxi); viii (Hebei); viii-ix (Guangdong); 7.viii (Zhejiang); ix (Shaanxi); 12-18.ix (Shanghai); x (Shandong). Taiwan: ix (Miaoli Hsien). South Korea: 21.vii. Japan: 4.v-3.xi (Ryukyu Archipelago); 13.vii-19.ix (Honshu); 17.vii (Shikoku). Russia: 16-26.viii (Primorskiy Krai).
There are one to two generations a year in northern China, with adults mainly in July and August (Yang, 1978); however, individuals can be found between June and September over most of central and southern China. It is probable that most of these are migrants, especially as this species is more common in the autumn. In Nepal, the moth appears as early as March (Haruta, 1992).
OVUM: Pale green, almost spherical (0.84 x 0.85mm), shiny and smooth.

LARVA: Full-fed 60--80mm, width 11mm, horn 8mm. According to Bell & Scott (1937), in the first instar pale yellowish-green, with a short straight black horn. In the second instar, head yellowish-green, body dark green; eye-spots on segments 5 to 11 black above, yellow below; horn short, black with base yellow. By the third instar, head and body slate-colour. There is a dorso-lateral line of yellow spots on 3 and 4 on a background of deep black. Eye-spots with a round yellow pupil edged broadly with black, decreasing in size backwards. Horn black, a yellow spot on each side of its base. In the fourth instar, head slate-colour, body black. There is a slaty-black saddle-shaped shield on segment 2, with a pale yellow dorso-lateral spot on the front edge; spots on 3 and 4 as in Third instar; eye-spot on 5 and 6 with a round black spot in the middle of the yellow, on 7 to 11 of a darker shade of yellow. Horn long, thin, straight, black with a white tip and a yellow ring near the base.






In the fifth and final instar, head small, dull and smooth. Body dull and smooth, tapering fist gently then more sharply forwards from 8 and gently backwards from 8; segments 4 and 5 not much swollen. Horn straight, of medium length, thin, nearly cylindrical, tip truncate with a minute, low tubercle at each lateral angle; surface shiny, covered with very minute tubercles (Bell & Scott, 1937).
Head black; labrum canary-yellow; ligula black; basal segment of antenna canary-yellow, other segments whitish; mandible black. Body velvety-blackish on segments 2 to 4, rest of body plumbeous with short black stripes around the secondary rings. There is a dorso-lateral line of spots on 2 to 4, some yellow, some orange, continuing as a stripe formed of small grey dots, interrupted by the eye-spots, to base of horn. A broad, soiled, pale yellow subspiracular stripe is also present from 2 to 12, dotted with small black rings on 6 to 12. Eye-spots on 5 and 6 with a round black pupil in which lie two narrow, irregularly concentric rings of electric blue; this pupil edged narrowly above, more broadly elsewhere with orange, at the top and bottom of which is a crescent of electric blue; the whole edged broadly with velvety-black. On 7 to 11 the eye-spots are rather larger, pupil deep purple above shading to reddish-brown below, edged above and below with a crescent of electric blue, the whole edged broadly with velvety-black; a broad yellow band, crossed by black lines, lying along the front margin of segments 5 to 11, and passing over the dorsum from the dorso-lateral stripe. Horn black with the tip narrowly yellow or white; legs red; protege and claspers black. Spiracles elongate-oval, white with a broad fuscous band across the middle, and a narrow black rim (Bell & Scott, 1937).
The thin horn is movable in a vertical plane in all instars.
The larvae are mainly diurnal and prefer younger leaves, seedpods and flowerheads, often stripping growing shoots, particularly in the final instar. Several of the gaudy larvae can often be found on one small plant.



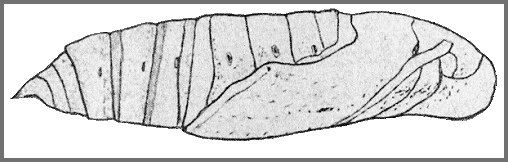
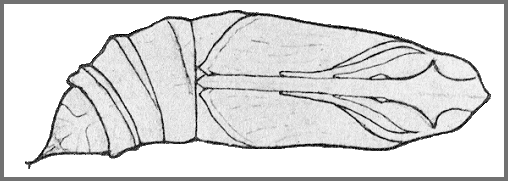
PUPA: 39--40mm, width 11mm. Colour pale yellowish-brown (darker and redder in overwintering individuals); legs dotted with black; abdomen dotted with black, these dots forming an obscure dorsal and similar latero-ventral stripe; a narrow interrupted ventral stripe; spiracles black, the ridge behind that on 2 dark brown, and the rims round the slits reddish-brown; cremaster dark reddish-brown, shaft and points black (Bell & Scott, 1937).
Tongue-sheath not much projecting (3mm frontad), semicircular in side-view, directed somewhat ventrad. Hind margin of segment 11 slightly undercut and overlapping the front margin of 12. Surface shiny, head, thorax and wing-case, nearly smooth, abdomen shagreened and minutely pitted; front bevels of segments 9 to 11 transversely corrugate. Spiracle of 2 a slit lying between the forward-curved, slightly, raised, hind margin of 2 and the straight front margin of 3, behind which is a short rounded transverse ridge; remaining spiracles oval, surface convex, central slit with narrow raised rim. Cremaster elongate-triangular, ending in a short, stout truncate-conical polished shaft, tip shaped like the tail of a whale, the flukes formed of short conical highly polished teeth directed outwards; upper surface convex and pitted, lower surface slightly hollowed, wrinkled, with a rounded mesial ridge from base to shaft (Bell & Scott, 1937).
In a loose cocoon amongst leaf litter on the ground.


Larval hostplants. Recorded in China on Arisaema, Cayratia, Cissus, Colocasia, Impatiens, Linum, Ludwigia, Pinellia, Typhonium and Vitis (Mell, 1922b; Yang, 1978; Chu & Wang, 1980; Pittaway, pers. obs. 2016). Li & Guo (1990) also give sweet potato (Ipomoea batatas) for Shanxi. A record of Humulus (Chu & Wang, 1980) is probably erroneous. The record on Carya (Juglandaceae) (Chu & Wang, 1980) is probably a transcriptional error for Careya (Lecythidaceae) from Bell & Scott (1937).
On Taiwan, recorded from Alocasia macrorrhiza, Ampelopsis brevipedunculata, Causonis japonica [syn. Cayratia japonica], Colocasia esculenta [syn. Colocasia formosana], Colocasia antiquorum, Epipremnum aureum, Leucocasia gigantea [syn. Colocasia gigantea], Oldenlandia diffusa [syn. Hedyotis diffusa], Impatiens balsamina, Impatiens walleriana, Ludwigia hyssopifolia, Ludwigia peploides, Ludwigia octovalvis, Oxalis debilis var. corymbosa [syn. Oxalis corymbosa], Pentas lanceolata, Typhonium roxburghii [syn. Typhonium divaricatum] and Typhonium blumei.
In Japan, recorded from Alocasia odora, Causonis japonica [syn. Cayratia japonica], Colocasia esculenta, Leucocasia gigantea [syn. Colocasia gigantea], Impatiens balsamina and Ipomoea batatas.
In the Russian Far East recorded from Vitis amurensis and Impatiens balsamina (Izerskiy, 1999b).
In Metro-Manila, the Philippines, larvae have been found on various species of Cayratia (Dvořák, 2014).
Outside of eastern Asia, the larvae of this species are not only an occasional pest of cultivated grapes (Goodwin, 1986) and sweet potato (Hamilton & Toffolon, 1986), but also feed on the ornamental bedding plant Impatiens wallerana in the gardens of eastern Australia (Herbison-Evans & Crossley, 1998), as well as Fuchsia. The larval hosts of Theretra oldenlandiae are listed for Australia by Moulds (1981), who also reports the larvae as occasionally defoliating commercial vineyards.
[Can be reared in captivity on lettuce (Lactuca sativa). If buying from a supermarket during the winter months, use organic produce (Bernie Betts, 2015).]
Ichneumonidae: Charops hersei Gupta & Maheshwary, 1971, Amblyjoppa cognatoria (Smith, 1874), Amblyjoppa proteus satanas (Kriechbaumer, 1895); Tachinidae: Exorista japonica (Townsend, 1909).
Charops hersei Gupta & Maheshwary was observed parasitizing larvae of Theretra oldenlandiae at Trivandrum, Kerala, India. The life cycle of the ichneumonid lasted 22-27 days, with field parasitization rates of 6-10% (Palaniswami and Pillai, 1983).
China: Heilongjiang; Liaoning; Jilin; Hebei; Shandong (Qingdao); Shanxi (Wenxi; Xiaxian; Yuncheng); Shaanxi (Xunyang, 1380m); Henan; Anhui (Mt. Huang Shan; Ma'anshan; Chizhou); Shanghai; Zhejiang (Hangzhou); Hubei (Hefeng; Mt. Guifeng, Macheng); Sichuan (Wolong National Nature Reserve; Pengshui); Yunnan (Laojun Shan, 2579m; Gaoligong Shan); south Xizang/Tibet (Zhangmu, 2200m; Yi'ong, 1900m); Hunan; Jiangxi; Guangdong (Guangzhou); Hong Kong; Guangxi; Hainan.
Taiwan: Miaoli Hsien; Taipei Hsien; Taipei (Neihu); Taoyuan Hsien; Kaohsiung Hsien; Taitung Hsien; Pintung Hsien; Hualien Hsien; Chiayi Hsien.
South Korea: Seoul; South Kyongsang Province (Pusan); Cheju Province; Uidong.
Japan: Hokkaido; Honshu (Tokyo; Nakabusa Spa; Kanetsu; Nashimoto; Tokyo; Oki Islands (Kadowaki & Kishida, 1977)); Shikoku (Kochi); Kyushu; Tanegashima; Ryukyu Archipelago (Yakushima; Amamioshima; Okinawa; Miyakojima).
Russia: Primorskiy Krai (Khasan; Primorskiy; Kedrovaya Pad nature reserve).
From Sri Lanka and southern India north to northern Pakistan (Rafi et al., 2014; Haxaire, Gujjar & Saeed, 2017), northern Afghanistan, Nepal, Sikkim - India (Khan & Raina, 2017; Chakraborty, Chakraborty, Biswas, Chakraborty & Deb, 2024), Bhutan (Irungbam & Irungbam, 2019), Madhya Pradesh - India (Chandra, Pandey, Bhandari & Sambath, 2013) and Myanmar/Burma. Thence northeastwards through China to Taiwan, South Korea and Japan, and then southeastwards through South East Asia as far as the Andaman and Nicobar Islands (Singh, Ahmad & Chandra, 2021), the Solomon Islands, New Guinea and the Philippines. Strongly migratory northward to northeastern China (Heilongjiang, Liaoning and Jilin), eastern Russia (Primorskiy Krai) and northern Japan; however, this species is only a rare vagrant to the Russian Far East (Derzhavets, 1984).
Reported from Lower Egypt by Abdelfattah Mabrouk Amer Salem (2022). This needs confirmation.
Most of continental Australia (especially the wetter north and east) as Theretra oldenlandiae lewini (Thon, 1828), although many taxonomists include this poorly differentiated taxon within the nominate race.
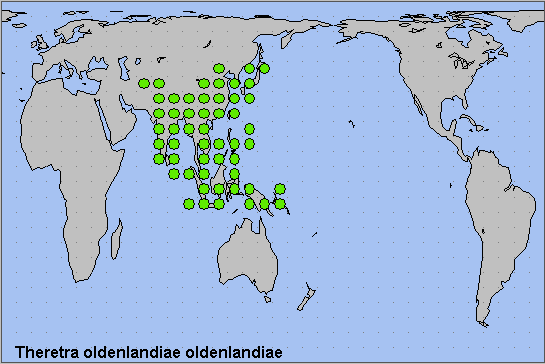
Oriental.
 Return to Sphingidae of the Eastern Palaearctic species list
Return to Sphingidae of the Eastern Palaearctic species list