

Sphinx alecto Linnaeus, 1758, Syst. Nat. (Edn 10) 1: 492. Type locality: India.
Synonym. Sphinx alecto Linnaeus, 1758.
Synonym. Sphinx cretica Boisduval, 1827, Annls Soc. linn. Paris 6: 118.
Synonym. Theretra freyeri Kirby, 1892, Synonymic Cat. Lepid. Heterocera 1: 650.
Synonym. Theretra alecto transcaspica O. Bang-Haas, 1927, Horae Macrolepidopt. Reg. palaearct. 1: 80.
Synonym. Theretro alecto intermissa Gehlen, 1941, Ent. Z., Frankf. a. Main 55: 185--186.
Note. The coloration of adults of this species can be influenced greatly by environmental conditions experienced during the larval and pupal stages. This can give rise to environmentally produced forms, such as 'subsp.' transcaspica, 'subsp.' cretica and 'subsp.' intermissa; none warrant subspecific status.
[Further details on this species in Japan, as well as photos of many stages, can be found on Digital Moths of Japan.]
Wingspan: 75--106mm. As depicted, with very little variation, apart from the intensity of coloration. Forewing upperside ground colour beige; fifth postmedian line, and sometimes the first line, heavy; lines two and four weak, lines six and seven vestigial or absent. Hindwing upperside red; basal area black, extending along inner margin towards the tornus. Immediately recognizable from all other Theretra (except Theretra mansoni) by the red hindwing upperside with a black base and the lack of a strong dorsal pale line along the thorax and abdomen. Labial palp segment one apical cavity not well defined proximally due to long scales projecting over it. Dorsal abdominal lines inconspicuous or absent. Outer row of forebasitarsal spines single, or partly double.
Specimens from more arid and desert habitats tend to be paler, whereas specimens from more humid forested habitats tend to be darker However, a number of forms exist: f. transcaspica O. Bang-Haas bears an orange-red, oblique, submarginal line on the forewing; f. cretica Boisduval is paler than normal, with a buff tint to the forewing and orange-red hindwing. These forms are geographical, but not exclusively: the former occurs in Turkmenistan; the latter seems to be found mainly in the drier and hotter regions of south-eastern Europe and the Middle East.
In the male genitalia, uncus rather narrow, slightly concave beneath apically, clothed with erect hairs, the apex rounded, weakly sinuate. Gnathos obtusely pointed, apex upcurved, strongly convex beneath. Valve with numerous stridulatory scales. Harpe slender, weakly sigmoid, apically pointed. Phallus apically with a short, multidentate process on the right, and a long oblique row of teeth on the left, on a slightly elevated ridge that ends in a slender process lying closely on the phallus. In the female genitalia, antevaginal plate with ridge proximal to the ostium bursae rather thin, but well sclerotised and smooth, becoming lower posteriorly to form a half-moon shape.



Occurs in areas where grapes are grown. Little is known about the behaviour of this species except that it is attracted to flowers and light.
In Metro-Manila, the Philippines, this synanthropic species has been reported as being active at daybreak and during rainy weather, when it has been observed to drink from water puddles next to roads (Dvořák, 2014).
China: 30.iii-1.vi (Hong Kong); 11.iv (Yunnan); v (Fujian; Yunnan; Guangdong; Hainan); 12.vi (Yunnan); 23.ix (Fujian); x (Fujian; Guangdong). Taiwan: vii (Chiayi Hsien); ix (Chiayi Hsien); x (Kaohsiung). Japan: 19.v-29.viii (Ryukyu Archipelago); 13.vi & 3.ix (Honshu); 28.vi-24.viii (Kyushu).
OVUM: Pale green, slightly oval (2mm), shiny and smooth; not unlike the egg of Laothoe populi. Up to five in a cluster may be laid on both the upper and lower surfaces of young leaves, each female depositing 150--250 eggs.
LARVA: Full-fed 80--110mm, width 11mm, horn 10 mm. Dimorphic: brown or green. According to Bell & Scott (1937), in the first instar pale yellow with a long straight black horn; the yellow body colour turns to green with feeding and this is the basic colour in the second instar; horn still black. In the third instar, head and body green, with a dark green dorsal stripe and white dorso-lateral stripe. Eye-spots are present in this dorso-lateral line on segments 5 to 11, that on 5 larger than the rest. These are either reddish or blue, ringed with black. Horn long, thin, base red or orange, rest black. (In this and the succeeding instars there is also a dark-coloured form of the larva.) In the fourth instar, green speckled with yellow, except on head and segments 2 to 5. There is a narrow dorsal stripe, black and sharply defined on 2 to 5, then brown and diffuse to 11. The dorso-lateral stripe is edged above by dark green shading. The eye-spot on 5 large and round, pupil black above, shading to red or brownish-purple, the pupil edged broadly below with bright yellow, narrowly above the white, and then with black. Remaining eye-spots longitudinally oval, pupil purple or red above, yellow below, the whole ringed with black. Horn of medium length, straight or curved gently up or down, base dull red, rest shiny black, sometimes with a white tip, the whole with self-coloured small tubercles.
In the fifth and final instar, head dull and smooth. True clypeus equilaterally triangular, about one-half length of head; false clypeus broadly arched over apex of true clypeus, reaching to slightly more than one-half length of head; labrum one-half length of clypeus, not quite as broad as clypeus, narrowing frontad; sinus very deep and rounded. Body dull and smooth, with segments 4 and 5 considerably swollen. Horn of medium length, stout at base, tapering evenly to a point, slightly down-curved; surface dull and covered with small tubercles (Bell & Scott, 1937).
In the green form, head grass-green; labrum and ligula green; antenna green, second and third segments tinged with pink; mandible green, tip reddish-brown, extreme tip black. Body dark green above the dorso-lateral stripe (pale yellow, from 2 to base of horn, interrupted by eye-spots), with short, broad, yellow stripes around each secondary ring of segments 6 to 11; paler green below the dorso-lateral stripe, closely dotted with whitish. There is also a narrow, dark green dorsal stripe. Eye-spot on 5 longitudinally oval, pupil with a black, pear-shaped marking at the top, below this green or purplish-brown, the pupil edged broadly below, narrowly elsewhere, with yellow of the same shade as the dorso-lateral stripe, the whole edged narrowly with brown or green. Eye-spots on 6 to 11 longitudinally elongate-oval, the upper half green or purplish, paler above, the lower half yellow, and contiguous with the dorso-lateral stripe, the whole edged narrowly with brown or green. Horn plum-colour or purple; legs red, the distal edge of each segment narrowly yellow. Spiracles lilac with a narrow brown rim (Bell & Scott, 1937).
In the dark form the green colour is replaced by olive-brown or brownish-pink; eye-spots as in the green form, but of a darker shade. There are seven broad dark brown oblique lateral stripes, the area round them pinkish.
In most full-grown larvae from tropical South East Asia, the dorso-lateral eye-spots are noticably larger than those found in middle eastern and european examples. It is not known whether this feature is determined by race or ecology, i.e. larger eye spots give a more pronounced startle response amids denser foliage. This feature was also noticed by Eitschberger & Ihle (2019).
It grows very rapidly and achieves its final length and a diameter of 10--12mm in 15--25 days. With its preference for young vine shoots and its abundance in certain localities, it can cause considerable damage. Having finished feeding, it descends very quickly from the hostplant in search of a suitable place to pupate.


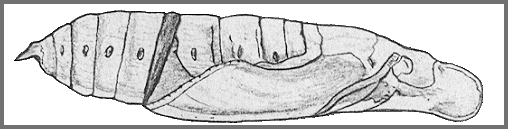
PUPA: 45--69mm, width 13mm. Colour dull ochreous; tongue-case reddish brown; head, abdomen with a very obscure greenish dorsal stripe; hind bevels of segments 8 to 11 chocolate, front bevels of 9 to 11 pinkish; spiracular and ventral regions speckled and striped with brown; spiracles and cremaster black (Bell & Scott, 1937).
Tongue-case projecting 8mm in front of head. Fore leg reaching to about the middle of wing-case, antenna slightly longer; mid-leg to about three-quarters the distance to tip of wing-case: there is a narrow coxal piece. The hind margin of segment 11 slightly undercut and overlapping the front margin of 12. Surface moderately shiny. Tongue-case with a narrow mesial channel, sides with wide radial corrugations; head and thorax coarsely, superficially shagreened, wing-case smooth. Abdomen more finely corrugate; segment 9 with about twelve ante-spiracular ridges, more or less parallel, but anastomosing in places; 10 and 11 with fewer and less prominent ridges; dorsum of 14 deeply pitted. Spiracle of 2 a narrow slit nearly covered by a transverse oblong lobe projecting from the front margin of 3, the front edge of the lobe raised, hind edge depressed; remaining spiracles oval, with very thick raised rims. Cremaster triangular, ending in two short, conical, divergent teeth, their bases touching; upper surface convex and longitudinally, irregularly ridged, lower surface with a broad mesial ridge and lateral extensor-ridges, the whole very rugose; segment 14 shallowly hollowed under base of cremaster (Bell & Scott, 1937).
Pupation may take place in a variety of sites: most larvae construct a loosely spun cocoon amongst dead leaves on the ground; others may pupate under stones or beneath the bark of a nearby tree, without forming a cocoon; during the summer months many pupate above ground, forming a cradle of vine leaves pulled together, usually in the notch of a stem. The pupa is not attached to the inside of the cocoon. This stage lasts from fifteen days to five months, depending on the climate. Overwinters as a pupa.
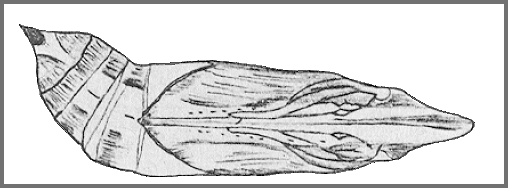

Larval hostplants. Recorded from Psychotria serpens in Macau (Easton & Pun, 1996) and Dillenia parviflora Griff. in Thailand (Eitschberger & Ihle, 2019). Elsewhere on Vitis, Parthenocissus, Cissus, Leea, Rubia, Psychotria, Saurauia, Dillenia and Tetracera.
On Taiwan from Ampelopsis brevipedunculata, Bischofia javanica, Causonis japonica [syn. Cayratia japonica], Euphorbia jolkinii [syn. Euphorbia formosana], Glochidion zeylanicum, Leea guineensis, Ludwigia octovalvis, Parthenocissus tricuspidata and Tetrastigma formosanum.
In Metro-Manila, the Philippines, larvae have been found on various species of Cayratia (Dvořák, 2014).
Unknown for the region.
China: Sichuan (Huili); Yunnan (nr. Yingjiang, Xima, 2080m; Pingbian; Gaoligong Shan; Wangjia; Menglunzhen; Xishuangbanna Tropical Botanic Garden); Xizang/Tibet (Mutu, Namjagbarwa region, 850m; Nyingchi/Linzhi, 3050m; Zhangmu, 2200m; Yi'ong, 2700m; Yadong, 2900m); Fujian (Xiamen); Guangdong (Guangzhou; Luofu Shan; Wanzishan, Deqing); Macau; Hong Kong; Hainan.
Taiwan: Chiayi Hsien (Tapang); Kaohsiung; Nantou Hsien (Puli); Pingtung Hsien (Koshun); Taipei Hsien; Taipei; Hualien Hsien (Hungyeh).
Japan: Honshu (Taira); Kyushu (Naze; Akaogi); Ryukyu Archipelago (Okinawa; Ishigaki-jima; Oshima-jima; Amamio-jima; Irabu-jima; Tokuno-jima; Iriomote-jima (Hayashi, 2017)).
From Greece and Bulgaria across southern and eastern Turkey to Transcaucasia, most of Iran, Turkmenistan, Uzbekistan, Kyrgyzstan, southern Kazakhstan and Afghanistan. From here south to Iraq, Lebanon, Israel and the more fertile areas of Egypt and Libya, and eastwards across Pakistan (Rafi et al., 2014), India (Chandra, Pandey, Bhandari & Sambath, 2013; Pathania, Sunita Sharma & Gill, 2014), Sri Lanka, Nepal, Bhutan (Dierl, 1975; Irungbam & Irungbam, 2019) and southern China to Taiwan, southern Japan, the Philippines and Indonesia.
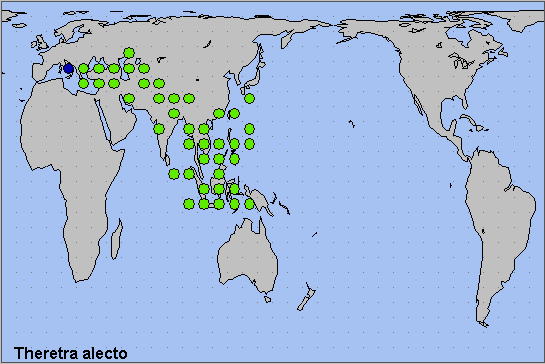
Palaeotropical; Oriental region. Penetrates the warmest areas of the Palaearctic region.
 Return to Sphingidae of the Eastern Palaearctic species list
Return to Sphingidae of the Eastern Palaearctic species list