

Sphinx nerii Linnaeus, 1758, Syst. Nat. (Edn 10) 1: 490. Type locality: not stated.
Synonym. Sphinx nerii Linnaeus, 1758.
Synonym. Daphnis nerii infernelutea Saalmüller, 1884.
Synonym. Daphnis nerii confluens Closs, 1912.
Synonym. Daphnis nerii nigra Schmidt, 1914.
Synonym. Deilephila nerii bipartita Gehlen, 1934, Bull. Mus. r. Hist. nat. Belg. 10: 2.
[Further details on this species in Japan, as well as photos of many stages, can be found on Digital Moths of Japan as well as Moths of the southern Shikoku, Japan.]
Wingspan: 84--126mm. Immediately distinguishable from all other species of Daphnis by the bright green upperside ground colour (rather than dark olive green or brown), but most easily confused with Daphnis hypothous (Cramer, 1780). Forewing upperside with antemedian line closer to basal patch than proximal edge of median green area. Hindbasitarsus considerably longer than tibia and also longer than tarsomeres 2-5; inner apical spur of hindtibia as long as hindtarsomeres 2-3. Variation confined to the intensity of wing coloration, especially the greens and pinks; most strikingly in f. nigra Schmidt, where green is replaced by black.
In the male genitalia, uncus long and strongly curved. Gnathos much shorter than uncus, broad at base and pointed at apex. Harpe with prominent curved sharp basal process and obtuse apical process, densely dentate dorsally. Phallus with a rounded process bearing short triangular left lobe pointing proximad and a short right lobe pointing horizontally.





Originally a species of dry river-beds, oases and warm hillsides with scattered oleander bushes; however, localities overgrown with this shrub tend to be shunned. In tropical South East Asia (and Oceania) it has come to favour the more open municipal parks, road margins and gardens where ornamental oleanders, Catharanthus roseus and Tabernaemontana are grown. In and around Metro-Manila, the Philippines, this synanthropic species is more common in the city suburbs than in rural areas (Dvořák, 2014). Dense forest is avoided.
Rests by day, either on a solid surface or suspended among foliage with which it blends; the head is tucked in, with the thorax and abdomen raised off the underlying substrate. Most emerge late in the evening but do not take flight until just before dawn, to feed avidly from such flowers as Nicotiana, Petunia, Lonicera, Saponaria and Mirabilis. Thereafter, flight periods are mainly just after dusk and before dawn. Under warm conditions, adults are extremely wary and, if disturbed, will take flight even during daylight hours. Pairing is a short affair usually lasting, at most, four hours but, occasionally, a couple will remain in copula until morning. Rarely found at light, unlike D. hypothous (I. J. Kitching, pers. obs.).
In Metro-Manila, the Philippines, reported as being active at daybreak and during rainy weather, when it has been observed to drink from water puddles next to roads (Dvořák, 2014).
China: 7.v (Hong Kong); vi-vii (Hunan); 11.vii (Yunnan); ix-x (Zhejiang); x (Hunan); x-ii (Hong Kong). Japan: v-xi (Ryukyu Archipelago, Okinawa); ix-xi (Kyushu); 23.xi (Shikoku).
Kendrick (2002) states that it is bivoltine in Hong Kong, occurring from October until February, with the occasional record in May.
OVUM: Light green, almost spherical (1.50 x 1.25mm), minutely pitted, small for the size of moth. Laid singly on both the upper and lower surfaces of young leaves of isolated, preferably sheltered, bushes, especially those at the foot of cliffs or near houses, or in clearings amongst trees. Females often fly around a plant several times before approaching with a pendulous flight. Most take up to twelve days to hatch but, during hot weather, some hatch in only five.

LARVA: Full-fed 90--130mm. Dimorphic: green or brown. Newly-hatched larvae (3--4mm), which consume their eggshells, are bright yellow with an unusually long, very thin, blackish horn. However, with feeding the yellow quickly assumes a greenish hue and, after the first moult, the primary colour becomes apple-green with a white dorso-lateral line from abdominal segment 1 to the horn, a small, white eye-spot on the third thoracic segment, black spiracles and pink true legs. With growth, the eye-spots become blue with white centres, ringed in black. The horn has an unusual bulbous 'cap' until the penultimate instar. Fully-grown larvae show little difference from younger ones, except for the change in eye-spots, and the horn losing its bulbous cap and becoming orange with a black tip, finely warted, and downward curved. In some individuals the dorsal surface is rosy, while in most the dorso-lateral line becomes edged in blue. In their final instar, some assume a bronze colour with rosy red anterior segments, which tends to mask the pre-pupation plum coloration; in all, however, the newly acquired blue-black dorsal pigmentation, the now black eye-spots and unchanged white spots on either side of the dorso-lateral line remain prominent. The true legs may become purple or even blue.
When young, larvae feed fully exposed on the topmost leaves and flowers; when larger, they tend to conceal themselves further down the branches, or even, when not feeding during daylight hours, on the ground under stones or leaf-litter. Those choosing to remain on the hostplant rest along the lower surface or stem of a leaf, with the first four segments of the body slightly hunched. When first disturbed the caterpillar stretches out to resemble an oleander leaf. With further disturbance, the anterior segments are arched up, suddenly revealing the startling eye-spots; at this point the noxious gut contents may also be regurgitated.






PUPA: 60--75mm. Colour of head, thorax, wing-cases and sides and venter of abdomen dull orange; dorsum of abdomen reddish-brown speckled with black. A narrow black stripe runs from the frons to the tip of the tongue, and there is a broader black dorsal stripe on segments 2 to 4. Spiracles also black, lying in large black patches; cremaster also black. Broadly rounded in front, the shoulders not prominent; segments 13 and 14 form together a short cone. A crescent-shaped mark partly encircles each eye. Antenna slightly shorter than fore leg, which reaches to middle of wing-case mid-leg reaching to three-quarters of the wing-case; a long, narrow coxal piece is present. Surface shiny, head, thorax and wing-cases smooth, abdomen coarsely pitted on dorsum, the pits forming irregular lines; venter transversely creased, segments 12 to 14 closely pitted all over. Spiracle of 2 a slit, with the front margin of 3 raised into a ridge behind it; remaining spiracles oval, convex, the central slit with raised edges. Cremaster short, thin, conical, ending in two short teeth, the surface rugose (Bell & Scott, 1937).
Formed in a loosely spun yellow cocoon among dry debris on the ground. The pupa is free in the cocoon moving the abdominal segments vigorously when touched. Rarely survives northern winters, but the main overwintering stage throughout its southern range.





Larval hostplants. Mainly the flowers and young leaves of the ornamental Nerium oleander and Tabernaemontana over most of its range (including Bangkok, Thailand). Also on Adenium obesum and Catharanthus roseus [syn. Vinca rosea] in Hong Kong (D. L. Mohn, pers. comm. 2002; Kent H. K. Li, pers. comm. 2002).
In Taiwan, recorded from Adenium obesum, Alstonia scholaris, Catharanthus roseus, Cerbera manghas, Nerium oleander, Rauvolfia verticillata [syn. Dissolaena verticillata], Tabernaemontana divaricata [syn. Ervatamia coronaria; syn. Tabernaemontana coronaria] and Tabernaemontana pandacaqui.
In Laos and Thailand, also recorded from Alstonia scholaris and Tabernaemontana divaricata (Eitschberger & Ihle, 2008). The latter has also been recorded from Pakistan (Muhammad Ather Rafi, pers. comm. 2014), the former from India (Dar, Jamal & Shah, 2022).
In Metro-Manila, the Philippines, larvae are common in the wet season on new growth of the roadside and garden ornamentals Adenium obesum, Catharanthus roseus and Nerium oleander. During the dry season various species of ornamental Gardenia and Tabernaemontana are preferred (Dvořák, 2014).
Tachinidae: Compsilura concinnata (Meigen, 1824), Exorista sorbillans (Wiedemann, 1830).
China: Zhejiang (Hangzhou; Zhoushan; Ningbo; Wenzhou); Yunnan (Xiaomengtong; Simao/Pu'er; Nujiang Lisu Autonomous Prefecture; Kunming; Xishuangbanna); Hunan (Hengyang; Changsha); Fujian (Fuzhou; Quanzhou; Xiamen; Kinmen; Zhangzhou); Guangdong (Jieyang; Heyuan; Guangzhou; Shenzhen; Dongguan; Zhuhai); Macau; Hong Kong (Tin Shui Wai Park; Yuen Long Park; North Point; Shau Ki Wan; Tseun Wan; Shek Kong; Lantau Island); Guangxi (Nanning; Beihai); Hainan (Haikou; Sanya).
Taiwan. Taipei Hsien (Taipei); Taichung Hsien; Nantou Hsien; Tainan Island; Kaohsiung Hsien (Pratas/Dongsha Island, South China Sea); Taitung Hsien; Pintung Hsien (Kenting National Park); Hsinchu Hsien.
South Korea. Jeju-do (Jeju.)
Japan. Honshu (Saitama); Shikoku (Kochi); Kyushu (Kagoshima; Naze; Fukuoka; Kirishima; Makurazaki); Ryukyu Archipelago (Okinawa, Amami-oshima, Aka-shima, Miyako-jima and Ishigaki-jima); Ogasawara/Bonin Islands.
This species was until recently only a rare vagrant to mainland China and Taiwan (Lin, 1987; Inoue, 1990). However, the population on Hong Kong appears to well established (Kent H. K. Li, pers. comm. 2002), and it has been picked up as a vagrant in Hangzhou and Ningbo, Zhejiang, during September and October. It has even become a pest of ornamental oleanders in Hunan (Lei & Lin, 2010).
Tropical Africa, North Africa and the Middle East to Afghanistan (Ebert, 1969) and Pakistan (Rafi et al., 2014). Thence eastward to tropical south-east Asia, including Borneo and the Philippines. As a migrant, it penetrates northwards into central Europe and central southern Asia. In 1960, this species established itself on Okinawa, Japan (Inoue, 1967; Inoue, 1974; Riotte, 1986); it has since moved farther north. In 1974 it arrived in Hawaii (Riotte, 1977; Beardsley, 1979; Riotte, 1986). It has also colonized the islands of Saipan and Guam (Moore & Miller, 2008), as well as Chichi-jima in the Ogasawara/Bonin Islands, Japan (Ohbayashi, 1998; Takeuchi & Ohbayashi, 2006).
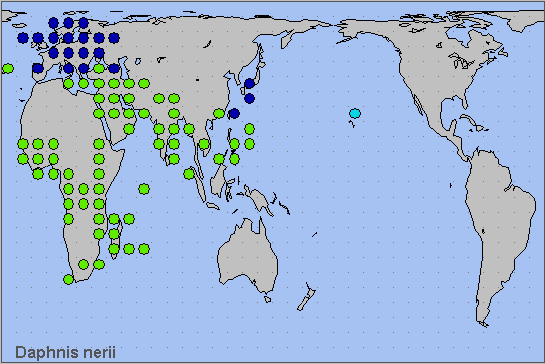
Palaeotropical.
 Return to Sphingidae of the Eastern Palaearctic species list
Return to Sphingidae of the Eastern Palaearctic species list