

Sphinx stellatarum Linnaeus, 1758, Syst. Nat. (Edn. 10) 1: 493. Type locality: not stated [Europe].
Synonym. Sphinx stellatarum Linnaeus, 1758.
Synonym. Sphinx flavida Retzius, 1783.
Synonym. Macroglossa nigra Cosmovici, 1892.
Synonym. Macroglossum stellatarum subnubila Schultz, 1904.
Synonym. Macroglossum stellatarum fasciata Rebel, 1910.
Synonym. Macroglossum stellatarum convergens Constantini, 1916.
Synonym. Macroglossum stellatarum approximata (Lempke, 1959).
Synonym. Macroglossum stellatarum clausa (Lempke, 1959).
Synonym. Macroglossum stellatarum candidum Eitschberger, 1971.
Synonym. Macroglossum stellatarum minor (Vilarrubia, 1974).
[Further details on this species in Japan, as well as photos of many stages, can be found on Digital Moths of Japan.]
Wingspan: 40--74mm. A very distinct species confusable only with some freshly emerged Hemaris species which have not yet lost their wing scales. Not prone to much variation though sometimes very pale, albinistic specimens occur, as do individuals with blackish brown abdomens and hindwings (f. subnubila Schultz). Antenna strongly clubbed, hook short, the terminal segment not very elongate.
In the male genitalia, uncus slender, gradually narrowed to a point, slightly hooked, not dilated apically either vertically of horizontally. Gnathos round at the tip. Valve without stridulatory scales. Harpe slightly curved, rounded-dilated apically, where it is rough with short spines and teeth. Phallus with one long, slender, pointed process, which is densely and heavily dentate on the proximal surface; the base of the process also heavily dentate, dilated distad. Internal rods obtuse, one clubbed and with a notched ridge; the other flat, concave on one side, with the edge finely serrate.


Diurnal. Whilst preferring to fly in bright sunlight, it will also take wing at dawn, at dusk or at night; in rain, or on cool, dull days. Very hot weather tends to induce a state of torpidity in many, with activity then confined to the relative cool of the morning and late afternoon. Whatever the flight-time, this species is very strongly attracted to flowers yielding plentiful supplies of nectar, such as Jasminum, Buddleja, Nicotiana, Tulipa, Primula, Viola, Syringa, Verbena, Echium, Phlox and Stachys, hovering in front of and repeatedly probing each bloom before darting rapidly to the next. A great wanderer, being present right across China from the alpine tree-line to city centres, wherever nectar flowers may be found. Its powers of flight are amazing, and have been studied in detail by Heinig (1987). Apparently, this species also has a fine memory, as individuals return to the same flower-beds every day at about the same time (Pittaway, 1993). (See also Heinig (1981a, 1984).)
When not feeding, pairs in courtship can be seen dashing up and down around steep cliffs, buildings, or over selected stretches of open ground. Pairs in copula can occasionally be found in such locations, although they seldom stay together for more than an hour. Whilst in copula, unlike other hawkmoths, this species is still capable of flight in a manner similar to butterflies. After a further period of feeding, gravid females search for patches of Galium growing in sunny locations. While hovering, each patch is carefully examined, sprig by sprig, before a single ovum is placed amongst the flower-buds. Up to 200 ova may be deposited by each female, therefore egg-laying can take a considerable time.
M. stellatarum is unique among sphingids of the region in overwintering as an adult, although in northern China very few survive. With the onset of cooler weather, individuals can be seen examining caves, rocky crags, empty houses, holes in trees or sheds, before selecting a suitable place for hibernation. This state of torpidity is not absolute, however, for warm days in December and January may bring some out to feed.
China: iii-vi (Zhejiang); iv (Jiangxi); iv-v (Shandong); v (Xinjiang, Yining/Gulja, 1000m); vii (Nei Mongol, Great Khingan Mountains, Zalantun/Butha Qi); 23.vii (Shaanxi); viii (S Gansu, 3100m); 4.viii (Zhejiang); ix (S Gansu, 2400m; Beijing); x (S Gansu, 1800m); x (Shandong; Heilongjiang; Hong Kong); x-xi (Liaoning); xi (S Gansu, 1550m); 29.xi (Hong Kong). Mongolia: 9-10.vi (Khovd Province); 11.vii (Gov'-Altai Province). South Korea: 11.vii (no locality). Japan: 20.ii-17.x (Hokkaido); vi (Shikoku); vi-x (Ryukyu Archipelago); viii (Honshu); 19.ix (Shikoku). Russia: 28.ii (Primorskiy Krai); 7.iii (Primorskiy Krai); 4.v (Primorskiy Krai); 16.vi (Siberia); 26.vii (Altai); 16.viii (Primorskiy Krai); 20-31.viii (Kurile Islands); 27.viii (Beijing); ix (Primorskiy Krai); 11-20.ix (Kurile Islands); 14.x (Primorskiy Krai); xii (Primorskiy Krai).
Park et al. (1999) give early April until late October as the flight period in Korea.
OVUM: Pale green, almost spherical (0.84 x 0.95mm), shiny, smooth, resembling unopened flower-buds of Galium. This stage lasts from six to eight days.
LARVA: Full-fed 45--50mm. Polymorphic.
Newly-hatched larvae are about 2--3mm in length, cylindrical and clear yellow. They eat copiously from the outset and so grow rapidly. In the second instar they assume their green coloration, covered with tiny yellow dots. A dark grey, cream-bordered, dorso-lateral line runs from head to horn, matched by an identical ventro-lateral band. The dorsal heart line is deep green; the tail horn purplish red with an orange tip. This pattern remains to the final instar, with only the predominant colour of the horn changing from reddish to blue with an orange tip. In some, however, the green body colour is entirely replaced by reddish brown, which effectively masks the final, pre-pupation, rich plum coloration.
In the final instar head small and round; horn straight, of medium length, stout at base, tapering evenly to a sharp point. Surface dull, a transverse row of small tubercles along each secondary ring.
Feeding takes place fully-exposed at the top of the plant, with no time preference being shown. When resting, or moulting, most larvae retire to within the bed of tangled stems. With ample food and sunshine, this stage may be as short as 20 days. (See also Heinig (1981a, 1984).)
Can be found throughout the summer months, with two peaks of abundance in the north: one during May and June, the other during August. Larvae are particularly common in the outskirts of Beijing in late August on Rubia clumps growing in unkempt shrubberies. In fact, occurs almost anywhere where Galium and Rubia are present in open situations, i.e. commons, urban parks, field edges, roadside verges, sand-dunes, railway embankments, and even high alpine meadows over its western range.



PUPA: 30--35mm. Pale translucent brownish cream, splashed with darker brown, especially on wings and spiracles. Proboscis prominent, keeled, laterally compressed, black along keel. Surface shiny, slightly shagreened. Cremaster terminating in two sharp spines. Enclosed in a silken cocoon spun low down among the hostplant, or among debris on the ground. Does not overwinter; however, Derzhavets (1984) claims that in Central Asia a proportion of pupae do overwinter.


Larval hostplants. Recorded in China on Stellaria media and Rubia cordifolia (Yang, 1978; Pittaway, pers. obs 2003); also Leptodermis oblonga (Zhang YuChen, pers. comm. 2023). Yang (1978) also listed Glycine max, while Chu & Wang (1980) added Panax pseudoginseng var. notoginseng; both records are erroneous. Elsewhere, Macroglossum stellatarum is recorded primarily on Rubiaceae and mostly species of Galium and Rubia.
Recorded in Korea on Rubia akane, Galium dahuricum and Galium verum var. asiaticum (Park et al., 1999). The first and the last species are also utilized in Japan.


Unknown for the region.
China: Xinjiang (ürümqi; Yining/Gulja); Nei Mongol (Zalantun/Butha Qi; Horinger; Alxa Zuoqi; Dujiahaocun, Togtoh County); Heilongjiang (Hailin; Lalin); Liaoning (Dalian); Hebei (Yangyuan; Zhengding); Beijing (Haidian; Ming Tombs; Xiangshan); Tianjin; Shandong (Jinan; Qingdao; Tai'an; Weihai; Yantai (Fauvel, 1876)); Shanxi (Taiyuan; Xiaxian; Yuncheng); Shaanxi (Huangling; Qinling); Gansu (Min Xian; Beishui Hu; Dangchang); Henan; Shanghai; Zhejiang (Hangzhou; Tian Shan; Mogan Shan); Sichuan (??Chin-chin-kai; Nanchuan); Xizang/Tibet (Mutu, Namjagbarwa region, 850m); Hunan; Jiangxi (Nanchang); Guangdong; Hong Kong.
Kendrick (2002; 2010) regards this species as a vagrant to Hong Kong, with only two individuals having been recorded.
Taiwan: A rare vagrant, with most individuals having been recorded around Taipei in the north.
Mongolia: Khovd Province (Arshantyn Nuruu Mts., Bulgan River valley, River Ulyastajn-Sala, 1900m (46°21'N 91°08'E)); Gov'-Altai Province (Mongolian Altai Mts. (south slope), Mogoijn River valley, 1800m).
North Korea: South Pyongan Province (Pyongyang); Kangwondo Province (Keumgang-san); South Hamgyong Province (Gyungsung); North Hamgyong Province (Jueul; Hoeryong; Samseong-san).
South Korea: Widespread in Seoul; Kyonggi Province; Kangwon Province; North Chungchong Province; South Chungchong Province; North Cholla Province; South Cholla Province; North Kyongsang Province; South Kyongsang Province; Cheju Province
Japan: Hokkaido (Hakodate); Honshu (Fujimi Heights; Kiyusato); Shikoku (Matsuyama; Tosashimizu); Kyushu; Tsushima; Ryukyu Archipelago (Okinawa; Ishigakishima (Kishida & Shirakawa, 1988)).
Russia: Altai (Cherga; Yailyu; Rebrikha village); Siberia (Novosibirsk area; Alaevo); Yakutia/Sakha (Yakutsk); Buryatia (Baikalskii Nature Reserve); Amurskaya (Blagoveshchensk; Uril area); Khabarovskiy Krai (Khabarovsk City); Primorskiy Krai (Jankowski Peninsula; Askold Island; Vladivostok; Khasan; Primorskiy; Kedrovaya Pad Nature Reserve); Kamchatka; Sakhalin Island; Kurile Islands (Kunashir Island).
Resident across the entire northern subtropics of the Old World, with migrants reaching far to the north during summer, including Yakutsk (Kaimuk et al., 2005), Kamchatka, and the Kurile Islands (Dubatolov, Zinchenko & Ustjuzhanin, 2023; Spitsyna & Spitsyn, 2023). Occurs as a migrant across all China (Yang, 1978), with the first adults appearing in Beijing in April.
Occasional specimens have been recorded in the tropics (e.g. Hong Kong, South India, Vietnam, Malaysia). These could be migrant individuals, but some may also be misidentifications of other Macroglossum species. A record from Congo-Brazzaville (Rougeot, 1972) was probably an accidental introduction, perhaps via an aeroplane. A single specimen from the University of Washington collection labelled 'Unimak Island' (in the eastern Aleutian Islands, Alaska) (M.D. van Buskirk, pers. comm.) explains the inclusion of Macroglossum stellatarum in the faunal list of the USA (Hodges, 1983). In fact, several specimens have been recorded from North America in the past. In 2017 a nectaring adult was photographed at Puente Hills (Whittier or Haceinda Heights), Los Angeles County, California, on April 17, 2017 (Ben Smith, pers. obs. 2017). These specimens probably arrived as hibernating adults in trade cargo from Japan or Europe. Since then this species appears to have firmly established itself as a breeding species in northern California and Oregon, USA, with several adventive adults noted elsewhere in the USA, even on the East Coast (Bill Farley, iNaturalist 2025).
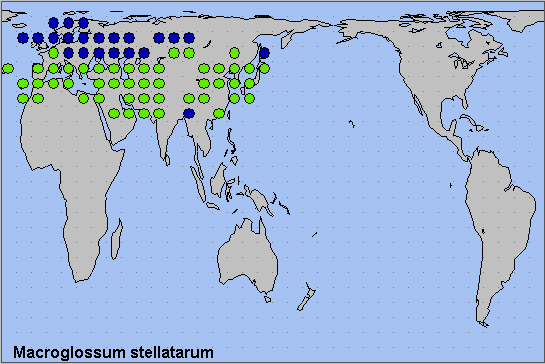
Holarctic; Palaearctic (both eastern and western subregions). Pleistocene refuge: Polycentric -- probably several refugia, from the Mediterranean to the Sinopacific.
 Return to Sphingidae of the Eastern Palaearctic species list
Return to Sphingidae of the Eastern Palaearctic species list