


Smerinthus carstanjeni Staudinger, 1887, In: Romanoff (ed.), Mém. Lépid. 3: 159. Type locality: [Russia, Khabarovskiy Krai/Primorskiy Krai] Ussuri; [Russia, Primorskiy Krai,] Suifun [Suifen].
Synonym. Smerinthus gaschkewitschii carstanjeni Staudinger, 1887.
Synonym. Marumba gaschkewitschii coreana Clark, 1937, Proceedings of the New England Zoological Club 16: 30.
Synonym. Marumba gaschkewitschii koreuemba Bryk, 1946, Arkiv för Zoologi (A) 38: 67.
Note. Marumba gaschkewitschii is currently divided into four subspecies: Marumba g. gaschkewitschii, Marumba g. complacens (Walker), Marumba g. carstanjeni (Staudinger), and Marumba g. gressitti Clark. However, the boundaries between these are not clear-cut, particularly within China. Specimens matching two or more subspecies can be found in the same locality at the same time, for example, as was observed at Yangling (17.vii.1995) and 30km north of Huangling, Shaanxi (24.vii.1995) (Pittaway & Kitching, 2000).
Three former subspecies/species, namely Marumba g. echephron (Boisduval, [1875]), Marumba harutai Eitschberger & Ihle, 2012, and Marumba g. irata Joicey & Kaye, 1917, are now considered to be distinct species (Eitschberger, 2012).



Marumba gaschkewitschii occurs in most habitats, including cities and orchards. Particularly common in montane forests rich in species of Prunus and Pyrus (Mell, 1935).
China: 8.vi-8.vii (Nei Mongol); 16-25.vi (Jilin); vi-vii (Heilongjiang); vii (Liaoning). North Korea: 1-12.vii (North Pyongan Province); vii-ix (Jueul, 1500m). South Korea: 20-28.vi (Ulleung-do); 22.vii (Jeju Do). Russia: v-28.vii (Primorskiy Krai); 14.vi-1.viii (Khabarovskiy Krai); 6-10.vii (Amurskaya).
Marumba gaschkewitschii s.l. has one to three generations a year. One is usual in Liaoning and Beijing, two in Ningxia, Hebei, Shandong, Jiangsu and Zhejiang; and three in Jiangxi (Chu et al., 1979). (Full-grown larvae were abundant around Beijing in late August 2003 (Pittaway, pers. obs. 2003).) Mell (1935) noted three to four generations (between 8.iii-5.x) in northern Guangdong and four to five generations (between 15.ii-2.xi) in southern Guangdong.
Park et al. (1999) give mid May until late August as the flight period in Korea.
OVUM: Oval (1.57 x 2.07mm). Initially a translucent pale jade green, but turning yellowish-green prior to hatching.

LARVA: Full-fed 75--83mm.
Newly hatched larvae are pale yellow with a round head and, in some, an orange tint to the horn. Only in the second instar does the characteristic triangular head appear, with the basic body colour turning gradually green. At this stage the final yellow oblique lateral lines and yellow body tubercles become more and more pronounced. The horn becomes more orange.




For the first few instars young larvae rest and feed stretched out along the midrib under a leaf. This behaviour makes them appear very elongate.
More mature larvae generally feed at between 0.5 and 1.5m off the ground on terminal branches of bushes rather than trees. Smaller plants set in amongst other shrubs are preferred. Larvae can be met with at very high densities, with often four or five per branch. Most larvae go through six instars.



PUPA: 40--48mm. Reddish mahogany brown and glossy; tapering caudad from a blunt head and thorax. Head tuberculate, with two vertical, blunt, broad crests frontad. Proboscis not present, but replaced by a knob-like tubercle. Wings smooth, abdominal segments finely punctate. Spiracular ridges present on movable abdominal segments. Cremaster broadly conical, with a sharp point; tuberculate. Similar to that of Callambulyx tatarinovii, but head more rough and with two crest-like tubercles frontad, as found in most species of Marumba. Formed in an almost silk-free cell in the soil. The overwintering stage.

Larval hostplants. Recorded in Primorskiy Krai, Russia, on Prunus, Pyrus, Crataegus (Derzhavets, 1984) and Sorbaria sorbifolia (Nikolay Ivshin, pers. comm. 2020); also on Malus baccata and Crataegus pinnatifida (Izerskiy, 1999b).

Recorded in Korea on Kerria japonica, Prunus mume, P. persica, P. salicina, P. serrulata, Malus pumila and Pyrus pyrifolia var. culta (Park et al., 1999). However, records from Buxus microphylla var. koreana are suspect.
For China, Yang (1978) and Chu & Wang (1980) also give Ziziphus mauritiana [Z. jujuba]. The first author was able to confirm this unusual hostplant by finding numerous full-grown larvae on Z. jujuba shrubs in the hills west of Beijing (Xiangshan) during late August (Pittaway, pers. obs. 2003). Larvae were also common on apricot Prunus armeniaca at Chengde. However, records from Buxus microphylla and Euonymus alatus (Chang, 1989) are suspect, while those on Vitis vinifera (Chu et al., 1979; Chu & Wang, 1980) are probably erroneous. The source and veracity of the record on Weigela (Zhang, 1994) are unknown.
In captivity the larvae found on Ziziphus and Prunus armeniaca readily transferred to Crataegus (Pittaway, pers. obs. 2003).
Unknown.
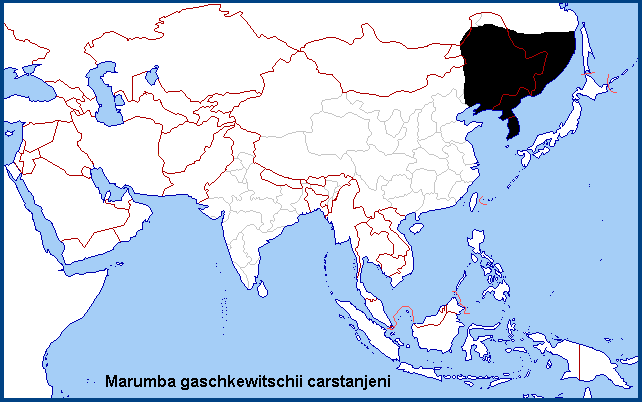
China: E Nei Mongol (Balin; Zalantun/Butha Qi; Chifeng/Ulanhad; Ongniud Qi; Uxin Qi; Hulunbuir Region, Sanhaodian); Heilongjiang (Harbin; Lalin); Jilin (Jiaohe, Lafa Shan); Liaoning (Huanren; Changhai, Dachangshan Island, Dalian); Beijing (Haidian); ?Hebei.
Korea: The entire Korean Peninsula (including Cheju-do and Ulleung-do).
Russia: SE Transbaikalia (Kyra; Khapcheranga; Undino-Posel'e); Amurskaya (Belogorsk; Blagoveshchensk; Uril area; Zeya); Yevreyskaya (Bastak Nature Reserve); Khabarovskiy Krai (Bolshekhekhtsyrskii Nature Reserve, Khabarovsk suburbs; Slavyanka; Komsomolsk-na-Amure; Pivan); Kamchatka; Primorskiy Krai (Kaymanovka; Lesogor'e; Dal'nerechensk; Vityaz Bay; Khasan area; Kedrovaya Pad Nature Reserve; Novovladimirovka; Pogranichniy; near Kalinovka; Andreevka; Anisimovka).
The core distributions of the current four subspecies of Marumba gaschkewitschii are as follows:
Three former subspecies, namely Marumba gaschkewitschii echephron (Boisduval, [1875]), Marumba gaschkewitschii harutai Eitschberger & Ihle, 2012, and Marumba gaschkewitschii irata Joicey & Kaye, 1917, are now considered to be distinct species (Eitschberger, 2012).
Southern Russian Far East, northeastern China and the Korean Peninsula.
 Return to Sphingidae of the Eastern Palaearctic species list
Return to Sphingidae of the Eastern Palaearctic species list